Answers
Answer:
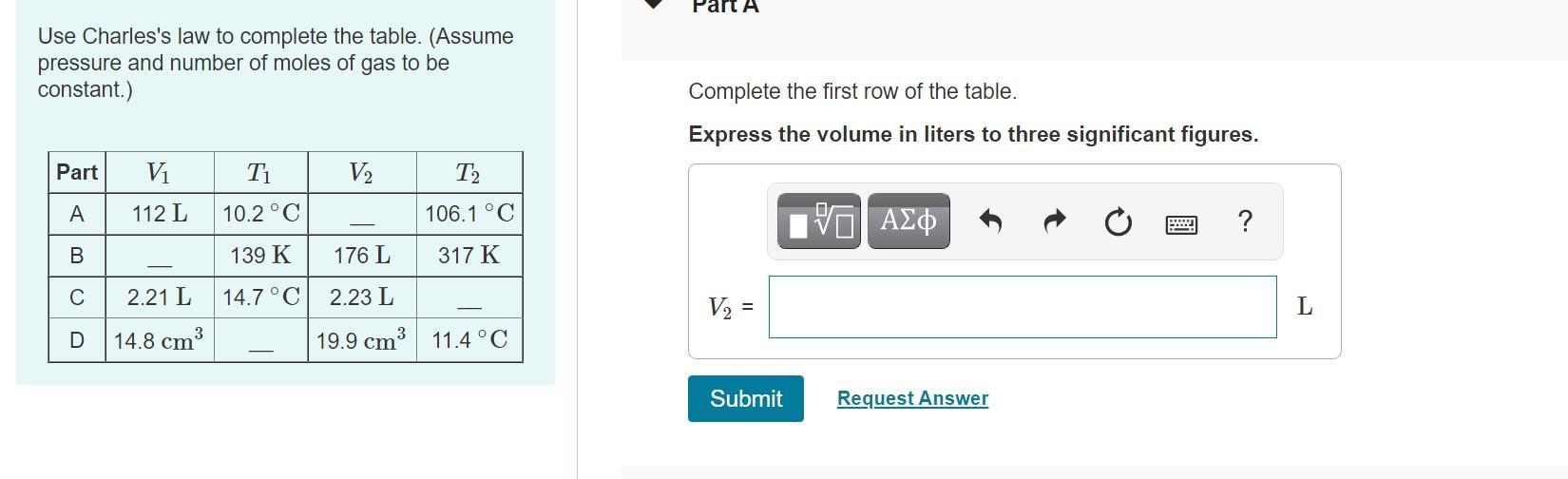
[tex]V_2\text{ = 149 L}[/tex]Explanation:
Here, we want to get the missing volume
From Charles' law, we know that volume and temperature are directly proportional at constant pressure
Mathematically, we know that:
[tex]\frac{V_1}{T_1}=\frac{V_2}{T_2}_{}[/tex]We have to rewrite the formula in terms of the missing value as follows:
[tex]V_2\text{ = }\frac{V_1T_2}{T_1}[/tex]Before we proceed to input the values, we have to convert the temperatures in celsius to kelvin by adding 273.15 K to the celsius temperature
Thus, we have it that:
[tex]\begin{gathered} V_1\text{ = 112L} \\ V_2\text{ = ?} \\ T_1\text{ = 10.2 + 273.15 = 283.35 K} \\ T_2\text{ = 106.1 + 273.15 = 379.25 K} \end{gathered}[/tex]We can now proceed to substitute these values into the rewritten formula as follows:
[tex]V_2\text{ = }\frac{112\times379.25}{283.35}\text{ = 149 L}[/tex]Related Questions
Can you describe the two different kinds of intermolecular forcesthat occur between diol molecules within a liquid?
Answers
A diol molecule is a compound that contains two Hydroxyl groups (OH), and these hydroxyl groups are attached to a hydrocarbon chain, which makes the main chain of the compound. In this type of molecule, we can find two intermolecular forces
Between the Carbons and Hydrogens in the hydrocarbon chain we have a Van der Waals force, which is a weak force
In the Hydroxyl groups, we will have a Hydrogen bond, which will be connecting Hydrogens and Oxygens
15. NI3 decomposes into Nitrogen gas and Iodine. If you start with 0.02 grams of NI3,How many moles of Nitrogen are produced at STP?
Answers
0.0000253moles
Explanations:The decomposition of NI3 is given as shown below;
[tex]2NI_3(s)→N_2(g)+3I_2(g)[/tex]Given the following parameters
Mass of NI3 = 0.02 grams
Determine the moles of NI3
[tex]\begin{gathered} moles\text{ of NI}_3=\frac{mass}{molar\text{ mass}} \\ moles\text{ of NI}_3=\frac{0.02}{394.719} \\ moles\text{ of NI}_3=0.0000507moles \end{gathered}[/tex]Acoording to stochiometry, 2 moles of NI3 preoduces 1 mole of nitrogen. The mole of nitrogen produced is;
[tex]\begin{gathered} mole\text{ of nitrogen}=\frac{1}{2}\times0.0000597 \\ mole\text{ of nitrogen}=\text{0.0000253moles} \end{gathered}[/tex]Hence the moles of nitrogen produced is 0.0000253moles
Find the formula mass of the compound, then divide the individual element total by the total mass-move the decimal over two to change it to percentage Al2O3
Answers
1) Find the formula mass of Al2O3.
Aluminum mass: 26.982 u.
Oxygen mass: 15.999 u.
Al2O3 mass = 2 * (26.982 u) + 3 * (15.999 u)
Al2O3 mass = 101.961 u.
2) Aluminum percentage.
Aluminum mass: 26.982 u.
Al2O3 mass = 101.961 u.
[tex]\frac{(2*26.982\text{ }u)}{101.961\text{ }u}=0.5293[/tex]Moving the decimal point.
52.93%
The percentage of aluminum is 52.93%
3) Oxygen percentage.
Oxygen mass: 15.999 u.
Al2O3 mass = 101.961 u.
[tex]\frac{(3*15.999\text{ }u)}{101.961\text{ }u}=0.4707[/tex]Moving the decimal point.
47.07%
The percentage of oxygen is 47.07%.
.
how to the chemical properties of sodium oxide allow them to be used
Answers
Answer:
Explanation:
Sodium oxide is an inorganic compound that has sodium and oxygen as its constituent
Majorly, its applications include in the ceramic industry and also aerounautical applications (for making light weight parts of air crafts)
So what exact properties of this oxide conform these properties?
The property here is that it is insoluble in water and other aqueous solutions
Furthermore, due to its ionic conductivity property, it finds use in applications like in the making of fuel cells.
In the following chemical wording, which atom gains electrons?(The formula is in the picture)The answers to choose are:•none•aluminum•oxygen•iron
Answers
Hierro
El número de oxidación del hierro en el lado izquierdo de la reacción es +3 mientras que en el lado derecho está como hierro metálico, es decir su número de oxidación es 0
How to draw (E)‑3‑methyl‑2‑pentene
Answers
In (E)‑3‑methyl‑2‑pentene, methyl group is attached in opposite side of the carbon that why it will be name as (E)‑3‑methyl‑2‑pentene.
The name of the given compound is (E)‑3‑methyl‑2‑pentene.
There are 5 carbon atom present in the compound. Methyl group is attached with C-2 and C-3 carbon atom in opposite direction
The structure of (E)‑3‑methyl‑2‑pentene is given as;
In the structure, it can be seen that methyl group is attached in opposite direction.
Trans molecule is named as E -compound.
To know more about Trans molecule
https://brainly.com/question/13981353
#SPJ1
Determine the following elements using their quantum #s of the elements last placed e-.include the e- configuration of the last placed electron.A. n = 4 l = 1, m = +1 , s = - ½,
Answers
If the question is asking us to find the element based on the quantum numbers of the last electron, we have these informations:
n = 4
l = 1
m = +1
s = -1/2
n represents the shell or level value, this value will generally go from 1 to 7, and since our value is 4, we are talking about an element whose valence shell is 4, so this element is in the 4th period
l represents the value of the orbital, this value can range from 0 to 3,
0 is the s orbital
1 is the p orbital
2 the d orbital
3 = f orbital
In out question we have l = 1, therefore the last electron of this element is in the p orbital and in the 4th period, now we are down to 6 elements, Gallium, Germanium, Arsenic, Selenium, Bromine and Krypton, all 6 elements are in the 4th period and have the p orbital as its last one
m represents the location of the electron within the orbitals, each orbital can hold 2 electrons only, p orbitals can hold 6 electrons in 3 pairs of 2, and the m value represents in which orbital the electron is, for l = 1, m can be -1, 0, +1
and in our question we have m = +1, therefore the electron is located in the last orbital, it can only be the 3rd electron or the 6th electron now, and now we are down to Arsenic and Krypton
s is the spin value of the electron, this value will tell us if the electron is pointing upwards or downwards in the orbital, this value can only be +1/2 (upwards) and -1/2 (downwards), and in our question, we have -1/2, which is the last electron of the orbital
Based on all that information, we conclude that this element is Krypton, with 36 of atomic number and electron configuration of [Ar]3d10 4s2 4p6
Which factors affect electronegativity?
A. Atomic radius and number of unshielded protons
B. Number of electrons and atomic radius
C. Number of unshielded electrons and density of the element
D. Density of the element and atomic radius
Answers
The factors that affect electronegativity is Number of electrons and atomic radius. Option A
What is electronegativity?The term electronegativity has to do with the the bonding situation between two atoms. In this situation, the electrons of the bond would be closer to one of the atoms than the other. In that case, there would be an unequal sharing of the electrons.
There are several facts that affect the electronegativity of an atom and these are the things that determine how electronegative that any particular atom would be.
Learn more about electronegativity:https://brainly.com/question/17762711
#SPJ1
Problem set3. What is the [OH-] of a solution that has a pH of 2?
Answers
We have a solution that pH = 2
We can use this relation which says:
pH + pOH = 14
If we clear pOH from here:
pOH = 14 - pH = 14 - 2 = 12
So, pOH = 12 and pOH could be calculated as:
pOH = - log [OH-] => we clear [OH-] from this,
10^(-pOH) = [OH-] => 10^(-12) = [OH-] => 1x10^-11 mol/L = [OH-]
(mol/L is generally the unit for [OH-])
Answer: [OH-] = 1x10^-11 mol/L
No clue on how to do these or how to show all my work and unit cancellations.
Answers
Question 16:
We are given the following balanced equation: (Remember to always balance the equation)
[tex]2HNO_3+Mg(OH)_2\rightarrow Mg(NO_3)_2+2H_2O[/tex]We are also told that Mg(OH)2 is in excess, meaning HNO3 is the limiting reactant, we will use its moles to find the number of moles of Mg(NO3)2, then we can convert that to mass.
The ratio between HNO3 and Mg(NO3)2 is 2:1
Therefore the number of moles of Mg(NO3)2 = 8.00 mol x (1/2) = 4.00 mol
Now we can convert the number of moles of Mg(NO3)2 to mass: molar mass of Mg(NO3)2 = 148,3 g/mol
[tex]\frac{8.00\text{ mol x 1}}{2}\text{ x }\frac{148.3\text{ g}}{1\text{ mol}}\text{ = 592 g}[/tex]So the mol will cancel the mol, then you will be left with g.
In the reaction 2 HgO --> 2 Hg + O2, how many moles of O2 are produced when 3.4 moles of HgO are decomposed?
Answers
In order to find the number of moles of O2 in this reaction we need to set up the properly balanced equation, which the question already gave us:
2 HgO -> 2 Hg + O2
From this equation we see that the molar ratio between HgO and O2 is 2:1, which means that for every 2 moles of HgO reacting we will end up having 1 mol of O2 as product, now what if we have 3.4 moles of HgO:
2 HgO = 1 O2
3.4 HgO = x O2
x = 1.7 moles of O2
How many moles are there for silver that are produced
Answers
0.96moles
Explanations:Given the reaction between Magnessium and silver nitrate expressed as:
[tex]Mg(s)+2AgNO_3(aq)\rightarrow Mg(NO_3)_2(aq)+2Ag(s)[/tex]Given the following parameter
Moles of Magnessium at the start = 0.480moles
According to stoichiometry, 1mole of magnessium produce 2 moles of silver. Hence the moles of silver that is produced if 0.48moles of Mg reacted is given as:
[tex]\begin{gathered} mole\text{ of Ag = 2}\times0.48moles \\ mole\text{ of Ag =}0.96moles \end{gathered}[/tex]Hence the moles of silver that will produced is 0.96moles
6. What would be the concentration of a solution formed when 1.00 g of NaCl are dissolved in water to make100 mL of solution?
Answers
To find the concentration we can do it in terms of molarity. Molarity is a way of expressing the concentration of a solute in a solution, it is expressed with the term M and can be described by the following equation:
[tex]Molarity=\frac{MolesSolute}{Lsolution}[/tex]So first we must find the moles of NaCl present in 1.00 grams, the moles of NaCl will be:
[tex]\begin{gathered} molNaCl=givengNaCl\times\frac{1molNaCl}{MolarMass,gNaCl} \\ molNaCl=1.00gNaCl\times\frac{1molNaCl}{58.44gNaCl}=0.017molNaCl \end{gathered}[/tex]So, the molarity of the solution will be:
[tex]Molarity=\frac{0.017molNaCl}{0.100Lsolution}=0.17M[/tex]Answer: The concentration of the solution in molarity will be 0.17M
Convert -4 degrees F to K.[?] KTin KelvinEnterQopyright © 2003-2022 dcelins Corporation. All Riss Reserved
Answers
From the question given, we are to convert -4⁰F to K
The standard value of ⁰F to K is
1⁰F = 255.93K
-4⁰F = x
Let's solve for -4⁰F
[tex]-4+255.93K=253.15K[/tex]From the calculations above, -4⁰F is equal to 253.15K
Hellllp pleaseeeee!!!
Answers
Answer:
A
Explanation:
The molecule is an amino acid which is used to make proteins, so it is an organic molecule.
It has a double bond between c=o
and single bonds throughout the molecule
Hope this helps!
If a block has the following dimensions and a density of 7,8 g/cm what is the masst?
L = 5.0 cm
W = 6.0 cm
H = 2.0 cm
Answers
The question requires us to calculate the mass of a block, given its density (7.8 g/cm~3) and dimensions (length = 5.0 cm; width = 6.0 cm and height = 2.0 cm).
Considering that the volume of the block can be calculated following the basic formula for volume (volume = length x width x height), we can obtain the volume of the object from the dimensions given and then use the definition of density to calculate the mass from the density and volume of the block.
First, we'll calculate the volume of the block:
[tex]\begin{gathered} \text{Volume = length}\times\text{ width }\times\text{ height} \\ \text{Volume = }(5.0cm)\times(6.0cm)\times(2.0cm) \\ \text{Volume = 60 }cm^3 \end{gathered}[/tex]Next, we must consider the definition of density (mass per volume of an object) to calculate the mass of the block:
[tex]\begin{gathered} \text{density = }\frac{mass}{\text{volume}}\to\text{mass = density}\times volume \\ \end{gathered}[/tex][tex]\begin{gathered} \text{mass = (7.8g/}cm^3)\times(60cm^3) \\ \text{mass = }468g \end{gathered}[/tex]Therefore, the mass of the block is 468g.
When 1.72 grams of copper metal react with 12.33 grams of silver nitrate how much silver is produced?
Answers
The silver produced is 7.77 g.
Given
Mass of Cu (copper) = 1.72 g.
Mass of AgNO3 (silver nitrate) = 12.33 g.
Molar mass of Cu = 63.5 g/mol.
Molar mass of AgNO3 = 170 g/mol.
Molar mass of Ag = 108 g/mol.
Step-by-step solution:
Cu + 2AgNO₃ --> Cu(NO₃)₂ + 2 Ag
First, let's state the chemical equation with copper (Cu) and silver nitrate (AgNO3) reacting:
1.72 g Cu . [tex]\frac{1 mol Cu}{63.5 g Cu}[/tex] = 0.027 moles of Cu
12.33 g AgNO₃ . [tex]\frac{1 mol AgNO_{3} }{170 g AgNO_{3} }[/tex] = 0.072 moles of Ag
Now, let's find the number of moles of each reactant using their molar mass:
0.027 moles Cu . [tex]\frac{2 moles Ag}{1 mol Cu}[/tex] = 0.054 moles Ag.
Now, let's see how many moles of Ag are being produced based on the number of moles that we found in each reactant. By doing this calculation, we will find the limiting reactant.
You can see in the chemical equation that 1 mol of Cu reacted, produces 2 moles of Ag, so the moles of Ag produced are:
And you can see that moles of AgNO₃ reacts, produces 2 moles of Ag too. This means that the molar ratio between them is 2, more simply sis 1:1. This is telling u that 0.072 moles of AgNO₃ will produce 0.0072 moles of Ag too.
In this case, as you can see the limiting reactant is AgNO₃ because this is being consumed first in the reaction, so the final step is to find the mass using its molar mass. The conversion from 0.0072 moles of Ag to mass in grams is:
0.072 mol Ag . [tex]\frac{108 g Ag}{1 mol Ag}[/tex] = 7.77
The silver that is produced by 0.0072 moles is 7.77 g of Ag (silver).
To learn more about moles, refer: https://brainly.com/question/26416088
#SPJ9
The Haber process for producing ammonia commercially is represented by the equation N2(g) + 3H2(g) → 2NH3(g). If 7 L of NH3 are consumed, how many liters of H2 gas are required?
Answers
Step 1
The reaction:
N2(g) + 3H2(g) → 2NH3(g) (balanced and completed)
All gases are assumed to be ideal and to be at STP conditions.
----------------
Step 2
STP conditions:
1 mole of gas = 22.4 L
----------------
Step 3
Information provided:
7 L of NH3 produced (ammonia)
Procedure:
By stoichiometry,
1 mole H2 = 22.4 L
1 mole NH3 = 22.4 L
N2(g) + 3H2(g) → 2NH3(g)
3 x 22.4 L H2 -------- 2 x 22.4 L NH3
X -------- 7 L NH3
X = 7 L NH3 x 3 x 22.4 L H2/2 x 22.4 L NH3 = 10.5 L
Answer: 10.5 L of H2 are consumed.
The atomic mass of copper (Cu) is 63 546 amu, the atomic mass of sulfur (S) is 32.065, and the atomic mass of oxygen (O) is 15.999 amu. Which molar mass is correct for copper sulfate (CuSO4)? (1 point) O 446.440 g/mol O 225.802 g/mol O 159.607 g/mol O 111.610 g/mol
Answers
The molar mass for copper sulfate (CuSO₄), given that the atomic mass of sulfur (S) is 32.065, and the atomic mass of oxygen (O) is 15.999 amu, is 159.607 g/mol
How do I determine the molar mass of CuSO₄?The molar mass of a compound is the sum of the individual atomic masses of the various elements that makes up the compound.
With the above information, we can obtain the molar mass for copper sulfate (CuSO₄) as follow:
Atomic mass of Copper (Cu) = 63.546 amuAtomic mass of sulfur (S) = 32.065 amuAtomic mass of oxygen (O) = 15.999 amuMolar mass of copper sulfate (CuSO₄) =?Molar mass of CuSO₄ = 63.546 + 32.065 + (4 × 15.999)
Molar mass of CuSO₄ = 63.546 + 32.065 + 63.996
Molar mass of CuSO₄ = 159.607 g/mol
Thus, we can conclude that the molar mass for copper sulfate (CuSO₄) is 159.607 g/mol
Learn more about molar mass:
https://brainly.com/question/18983376
#SPJ1
Answer: 159.607 g/mol
Explanation:
what is the water cycleA process by which water moves from cloud in the skyB A process by which water is absorbed into soil on earth surfaceC A process by which water moves from earth surface into the sky and back againD A process by which wind washes large amounts of water onto land from the oceans
Answers
Since water cycle is a multi-process cycle, the best option that describes and summarize all the cycle, will be letter C, in which we have water moving from the surface of the Earth to the sky and back again as rain, and then back to the sky again as water vapor.
Answer:
(C) A process by which water moves from Earth's surface into the sky and back again
Explanation:
hope this helps! <3
Perform the following operationand express the answer inscientific notation.7.00x105 – 5.00x104-[ ? ]x10[?]Coefficient (green)Exponent (yellow)Enter
Answers
The answer is 6.5x10^5
Coefficient = 6.5
Exponent = 5
Which one is not an organic coumpounds
Answers
Answer:
A
Explanation:
There are many definitions, and all of them are organic under some definition. The answer is probably A because it is the only one without hydrogen, and sometimes molecules without hydrogen are counted as being inorganic. A is CCl2F2, which is comprised of two chlorine atoms, two fluorine atoms, and a carbon atom.
How would this need to be answered? I don’t know how to do any of this.
Answers
Step 1
It is known that the rate law can be written as follows:
Given the reaction: X + Y => Products (completed and balanced), the rate of the reaction would be:
[tex]rate\text{ = k }\lbrack X\rbrack^a\lbrack Y\rbrack^b[/tex]k = rate constant
[X] = concentration of reactant X
[Y] = concentration of reactant Y
a, b = are the order of the reaction with respect to X and Y (they are not necessarily the coefficients of the reaction)
---------
Step 2
Information provided:
k
First order in A
Zero order in B
Second order in C
Therefore, the rate law:
Answer:
[tex]\begin{gathered} rate\text{ =k x }\lbrack A\rbrack^1x\text{ }\lbrack B\rbrack^0x\text{ }\lbrack C\rbrack^2 \\ rate\text{ = k x }\lbrack A\rbrack\text{ x }\lbrack C\rbrack^2 \end{gathered}[/tex]19.When a compound is added to water only a few of its molecules dissociate to produce hydrogen ions. It is a...Select one:a. strong acid.b. weak acid.c. strong base.d. weak base.
Answers
ANSWER
Weak acid ------ option B
EXPLANATION
Acid is divided in strong and weak acid
Generally, acid can be defined as a substance that will produce hydrogen ions as the only positive ions when dissolved in water.
Strong acid will dissolved completely in water to give hydrogen ions. Example of a strong acid is Hydrogen
Weak acid will dissolved partially in water to give hydrogen ions
Hence, a compound that will give only few of its molecules to produce hydrogen ions when dissolved in water is called a weak acid
Therefore, the correct answer is weak acid
Which statement is true concerning the structure and its functional group?Group of answer choicesCH3CH2OH is a caboxylic acidCH3CH2NH2 is an amineCH3CH2COOH is an esterCH3CH2SH is an ethernone of the above
Answers
Amine:
Amines are molecules that contain carbon-nitrogen bonds. The nitrogen atom in an amine has a lone pair of electrons and three bonds to other atoms, either carbon or hydrogen.
Functional group: NH2-R primary amine; NH-R secondary amine; N-R tertiary amine.
CH3CH2NH2 is the only one true. This is ethylamine
CH3CH2OH: OH- alcohol group
CH3CH2COOH: -COOH the functional group is a carboxyl group from a carboxylic acid.
CH3CH2SH: SH- this functional group is referred to as either a thiol group or a sulfhydryl group.
Answer: CH3CH2NH2 is an amine
the following unbalanced equation is performed in a laboratory:Mg + HCl --> MgClz + H2If you begin with 10.0 grams of each reactant, how many grams of hydrogen gas should you expect to measure in your experiment?
Answers
Answer: 0.276g of H2 could be obtained from the reaction, considering the mass of reactants given
Explanation:
The question requires us to determine the amount of hydrogen gas (H2) that would be obtained when 10.0g of metallic magnesium (Mg) and 10.0g of chloridric acid (HCl) are reacted.
The following unbalanced chemical equation was provided:
[tex]Mg+HCl\rightarrow MgCl_2+H_2[/tex]To solve this problem, we'll need:
1) obtain the balanced chemical equation;
2) determine the limiting reactant considering the amount of reactants used and the stoichiometry of the reaction;
3) calculate the mass of hydrogen gas produced, considering the limiting reactant
1) Balancing the chemical equation
From the unbalaced chemical equation, we can see that there is the same amount of Mg on both sides of the equation, but we need to adjust the amount of H and Cl atoms.
There are 2 Cl atoms and 2 H atoms on the right side, and 1 Cl and 1 H atom on the left side, thus we can adjust the coefficient of HCl from 1 to 2 to adjust these elements. The balanced chemical equation can be written as:
[tex]Mg+2HCl\rightarrow MgCl_2+H_2[/tex]2) Determining the limiting reactant
Now that we know the balanced chemical equation, we can say that 2 moles of HCl are necessary to react with 1 mol of Mg. Thus, we can write:
1 mol Mg ----------------------- 2 mol HCl
From this stoichiometric relation, we can calculate the amount of HCl that would be necessary to react with 10.0 g of Mg.
First, let's determine the amount of moles of Mg contained in 10.0g of this metal and the number of moles of HCl in 10.0g of this acid:
[tex]\begin{gathered} number\text{ of moles = }\frac{mass\text{ of sample \lparen g\rparen}}{molar\text{ mass \lparen g/mol\rparen}} \\ \\ number\text{ of moles of Mg = }\frac{10.0g}{24.31g/mol}=0.411mol\text{ of Mg} \\ \\ number\text{ of moles of HCl = }\frac{10.0g}{36.46g/mol}=0.274mol\text{ of HCl} \end{gathered}[/tex](the atomic mass of Mg is 24.31 amu, which is numerically identical to its molar mass, 24.31 g/mol, and the molar mass of HCl is 36.4g g/mol)
Therefore, 0.411 moles of Mg and 0.274 moles of HCl were used in the reaction.
Next, we can determine how many moles of HCl would be necessary to react with 0.411 moles of Mg:
1 mol Mg ----------------------- 2 mol HCl
0.411 mol Mg ----------------- x
Solving for x, we have that 0.822 moles of HCl would be necessary to react with the given amount of Mg. Since the amount actually used of HCl (0.274 mol) is smaller than the necessary amount, we can say that HCl is the limiting reactant.
3) Calculating the mass of H2 obtained
Using the limiting reactant and the stoichiometry of the reaction, we can determine the amount of H2 that could be produced.
From the balanced chemical equation, we know that 2 moles of HCl are necessary to produce 1 mol of H2. Thus, we can write:
2 mol HCl ------------------- 1 mol H2
0.274 mol HCl ------------ y
Solving for y, we have that 0.137 moles of H2 would be obtained.
We can convert the number of moles of H2 to its correspondent mass as it follows (the molar mass of H2 is 2.016 g/mol):
[tex]\begin{gathered} number\text{ of moles = }\frac{mass\text{ of sample \lparen g\rparen}}{molar\text{ mass \lparen g/mol\rparen}}\rightarrow mass\text{ of sample = number of moles}\times molar\text{ mass} \\ \\ mass\text{ of H}_2=0.137mol\times2.016g/mol=0.276g \end{gathered}[/tex]Therefore, 0.276g of H2 could be obtained from the reaction, considering the mass of reactants given.
Hello , can I have help with number 43 , please? Can you give explanation for all the choices ? Tank you
Answers
Number 43:
Please look at the next drawing:
Answer: b. upper right
Hey I miss my dad but I’m watching it and
Answers
Firstly what you need to know is that metallic bonds form in metallic substances. The elements that form these bonds are found on the left of the periodic table. So the metallic bonds form when the charge when the charge is spread over a wide distance when comparing to the size of the atom. These bonds occur between metals like zinc, and since they are in a form of solids, their atoms are tightly packed together.
The formation of metallic bonds occur because the metals exchange electrons, this electron exchange occur in different spatial directions.
A balloon originally has 0.100 moles of helium and has a volume of 0.500 L. If 0.530 grams of He are added to the balloon, what will the new volume be, in L?
Answers
The new volume of the balloon if 0.530 grams of He are added to the balloon is 1.16 L
What is the number of moles of helium added?The number of moles of helium added is determined as follows:
The final volume of the balloon if 0.20 moles of gas are added will be 1.62 L.
Moles = mass / molar mas
molar mass of helium = 4 g/mol
Moles fo helium added = 0.530 / 4
moles of helium added = 0.1325 moles
The final volume is determined using the ideal gas equation as follows:
PV = nRT
Where;
P is pressureV is volumen is the number of moles of gasR is molar gas constant = 0.082 atm.L/K.molInitial volume, V₁ = 0.50 L
Initial mole of gas, n₁ = 2.50 mol
With pressure and temperature held constant:
Final moles of gas, n₂ = 0.100 + 0.1325
n₂ = 0.2325 mols
Final volume, V₂ = n₂ * V₁ / n₁
V₂ = 0.2325 * 0.5 / 0.1
V₂ = 1.16 L
Learn more about gas volume at: https://brainly.com/question/27100414
#SPJ1
how many significant figures do the following numbers have?1) 5.9 x 104
Answers
Significant figures correspond to the number of digits that a number contains. Zeros at the beginning and end of the number are not counted, only zeros are counted if they are in an intermediate position.
For this case, the number is written in scientific notation, the corresponding 10 of the scientific notation is not taken into account during the digit count, therefore the significant figures of this number will be:
Answer: In 5.9 x10^4 there are 2 significant figures.