PLS HELP URGENT
Electron dot diagrams
Use your periodic table to write the electron dot diagrams for the following atoms.
1. Calcium (Ca)
2. Polonium (Po)
3. Moscovium (Mc)
4. Boron (B)
5. Fluorine (F)
Answers
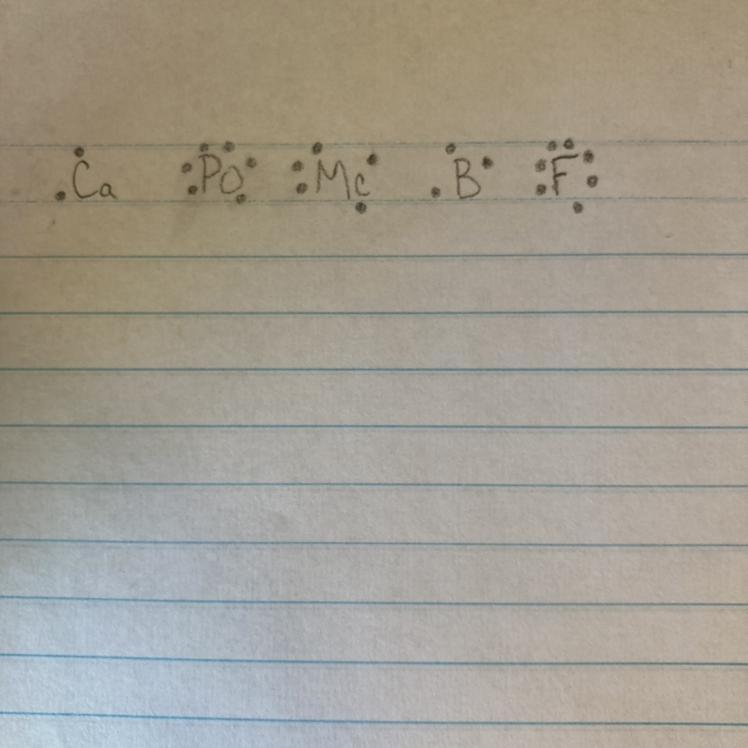
Related Questions
A compound and oxidant react to produce heat and a new product is what reaction
Answers
Answer:
A compound and oxidant react to produce heat and new product is chemical reaction.
PLEASE HELP ME.!
This illustrates which stage of meiosis?
A) Anaphase II
B) Metaphase II
C) Prophase II
D) Telophase II
Answers
Answer:
Anaphase II
Explanation:
For a theoretical yield of 5.52 g and percent
yield of 51.7877%, calculate the actual vield
for a chemical reaction.
Answer in units of g.
Answers
Answer:
2.85868104g
Explanation:
Actual yield
Theoretical yield x 100 = 51.7877%
So, do the inverse and you get 2.85868104g
PLZ HELP WILL BRAINLIEST balance the following equation: N2 + H2 --> NH3
Write the coefficients that you decide to balance the equation like this 3, 4, 3. If you do not add a coefficient in front of an element or compound, use a 1 in your answer. For instance the for this balanced equation: 2H2 + O2 --> 2H2O you would write your answer: 2, 1, 2
Answers
Answer:
1,3,2
Explanation:
N2 + 3H2-----> 2NH3
Four major bahiagrass varieties
Answers
Please help me, I am confused
Answers
Answer:
B
Explanation:
Ionic compound can conduct electricity
What would happen to a plant that did not perform cellular respiration in the light?
Answers
Answer:
Explanation:
Cellular respiration does not depend on light... Some plants may go into a form of hibernation during winter when the amount of light during the day is less. The reduction of plant growth and activity would reduce the need for the energy produced by cellular respiration.
what is atomic number of sodium
Answers
Answer:
Explanation:
Sodium is a chemical element with the symbol Na and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable isotope is ²³Na. The free metal does not occur in nature, and must be prepared from compounds.
Answer: Atomic number for sodium is 11
Symbol: Na
Melting point: 207.90°F (97.72°C)
The electronic configuration of sodium is 2, 8, 1
Do all mutations have the same affect on a cells dna
Answers
Answer:
no, some mutations can create cancers while others can create other infections
Explanation:
PLS HELP URGENT
Electron dot diagrams
Use your periodic table to write the electron dot diagrams for the following atoms.
1. Calcium (Ca)
2. Polonium (Po)
3. Moscovium (Mc)
4. Boron (B)
5. Fluorine (F)
Answers
What holds more salt warm or cold water?
Answers
13. If a chemist has 12.3 moles of N H 03, what is the mass of the sample?
Answers
Answer:
209.4831 g
Explanation:
number of moles × molar mass = mass of substance in g
12.3 × ( 14.0067 + 1.00484 × 3 ) = 209.4831 g
Calculate the number of moles of NaOH contained in 250. mL of a 0.05M solution?
Answers
Answer:
0.0125 moles of NaOH are present
Explanation:
Molarity, M, is an unit of concentration widely used in chemistry. Is defined as the ratio between moles of solute (In this case, NaOH) and liters of solution.
250.0mL are = 0.250L of solution. As the molarity of the solution is 0.05M = 0.05moles / 1L, the moles present are:
0.250L * (0.05moles / 1L) =
0.0125 moles of NaOH are present135 g of aluminum initially at 400.°C is added to an unknown mass of water (initially at 25°C). When thermal equilibrium is reached, the system has a temperature of 80.°C. Find the mass of water. Note c = .89 J/g°C for Al
Answers
Answer:
981.82g or 0.982 kg
Explanation:
I believe this is Physics, but okay!
We have to use the Specific Heat Capacity formula here.
Heat energy = mass x specific heat capacity x change in temperature
mass = Heat energy/(specific heat capacity x change in temperature)
Heat energy is measured in Joules so we need to find out how to do this.
To find an amount in Joules, we are given 135g and 0.89J/g°C.
135g x 400°C = 54000 to give us g°C
54000 x 0.89 = 48060 By multiplying these two values, we get given 48060J.
This is because: g°C/1 x J/g°C = Joules on it's own
mass = (48060)/[0.89 x (80 - 25)]
mass = 981.82g
"Don't use such phrases here, not cool! It hurts our feelings :( "
IS THE COMMENT EVERY DANG TIME YOU TRY TO TYPE ANYTHING TO POST! anyone else having this problem?
Answers
Answer:
ye, i always have that problem. my stuff gets deleted too :'((
What is the main function of the cell membrane in an animal cell?
Answers
Answer:
A cell's plasma membrane, also known as the cell membrane, provides protection. It also maintains a constant atmosphere within the cell, and the membrane serves a variety of purposes. The first is to transport nutrients into the cell, and the second is to transport toxic compounds out
Explanation:
Please help!!! What is electrolysis?
Answers
Explanation:
plzz tell me the ans even i also want to know plzzzz someone say ans
Answer:
electrolysis is a procedure that uses direct electric current to achieve an otherwise non-spontaneous chemical reaction and is important in the separation of elements from naturally occurring
How many moles are in 281 g of Ca(OH)2?
Answers
Answer:
3.79 moles
Explanation:
To convert moles to gams of a substance we need to find the molar mass of the substance. For Ca(OH)₂ th molar mass is:
1Ca = 40.08g/mol
2O = 2*16g/mol = 32g/mol
2H = 2*1.01g/mol = 2.02g/mol
The molar mass is:
40.08g/mol + 32g/mol + 2.02g/mol = 74.1g/mol
And moles are:
281g * (1mol / 74.1g) =
3.79 molesHumans become part of the carbon cycle when they
A. breathe in carbon dioxide,
B. release carbon as a waste product,
C. produce new carbon compounds in their cells,
Answers
Answer:
Human activities have a tremendous impact on the carbon cycle
Explanation:
Burning fossil fuels, changing land use, and using limestone to make concrete all transfer significant quantities of carbon into the atmosphere. ... The ocean absorbs much of the carbon dioxide that is released from burning fossil fuels.
When magnesium reacts with sulfuric acid the products are magnesium sulfate and hydrogen. If there are 15 grams of magnesium at the start of the reaction, how much magnesium will be present in the magnesium sulfate
Answers
Answer: 15 grams of magnesium will be present in the magnesium sulfate
Explanation:
According to the law of conservation of mass, mass can neither be created nor be destroyed. Thus the mass of products has to be equal to the mass of reactants. The number of atoms of each element has to be same on reactant and product side.
The balanced chemical reaction is:
[tex]Mg+H_2SO_4\rightarrow MgSO_4+H_2[/tex]
As the mass remains conserved at the end of the reaction, there will be 15 grams of Magnesium in magnesium sulphate.
If the mass of all the reactants in a chemical reaction is 100g, what will the mass of all products be?
Answers
Molar Volume of a gas at STP=22.4 L Example 3: Determine the volume of Carbon dioxide created when 15.0 grams of NaHCO3 are decomposed into water, Sodium carbonate, and Carbon dioxide at STP?
Answers
Answer:
20009 is answer is right 1345678899444
Explanation:
thhjbfthvcdthvctyhgffdy
gvhjk ghkde ghjcddxxb hhj hhgddxb ggjbcxdss ggbbsrtyg fy gbsdgh
How to convert celcius to Kelvin?
Answers
Answer:
temperature
Explanation:
What is the pOH if the [OH-]= 0.165 M? What is the pH of this basic solution? *Please round your answer(s) to the appropriate number of significant figures. Your answer can be in standard notation or i letter "e" in place of x10.* 1 N
Answers
Answer:
78
Explanation:
Answer:
The OH would be .7825 and the basic solution is a strong base.
Explanation:
What you would do is -log(0.165) in your calculator which would give you 0.7825160065 as an answer. Im not sure what the significant figure is so I will not be rounding to that, but that is your answer for the first part.
The second part: because your pOH is a .7825, this would be consiered a strong base in the pOH because it is closer to 1 which is your base.
what fault block that is located below the fault
Answers
Answer:
it is called the foot block
Which statement does NOT describe a relationship shown in the food web?
A Elk are prey for mountain lions.
B Mice are herbivores that consume grasses and are preyed on by snakes.
COwls prey on rabbits and frogs.
Rabbits consume shrubs and are parasites of grasses.
D
PLEASE HELP
Answers
I know this because rabbits aren’t parasites
What element is steel mainly composed of?
A. Iron
B. Carbon
C. Manganese
D. Silver
Answers
Hope this helps :)
El acero esta compuesto por Hierro y Carbono
The cells of all organisms must produce energy in order for the cell to survive and function. The diagrams below show
different parts of an animal cell and a plant cell.
In which organelle is energy produced in both plant and animal cells?
O Ribosomes
Nucleus
Chloroplast
O Mitochondria
Answers
Answer:
Mitochondria Is produced by both animal and plant cells
Explanation:
0.32 moles of oxygen gas has a temperature of 27°C and pressure of 2 atm in a closed container. What is the volume?
Answers
Answer:
3.9 L
First convert temperature to Kelvin
then use the ideal gas law
use algebra to solve for V
Since your solving for volume, your answer should be in Liters.
Sedimentary
Weathering
erosion, transport
deposition
Heat and/or
pressure
Igneous
Metamorphic
The
Rock
Cycle
Intrusion or
eruption
Burial and
extreme heat
Magma
Look at the diagram above.
Which sentence BEST describes a process in the rock cycle?
Igneous rocks can become metamorphic and sedimentary rocks.
Metamorphic rocks can form by the weathering process,
Volcanoes can deposit sedimentary rock to form mountains.
O Sedimentary rocks can melt to become metamorphic rocks.
Answers
Answer:
I am confident that it is the last answer but if you can find a more verified answer I would go for that.