HELP ASAP PLEASE
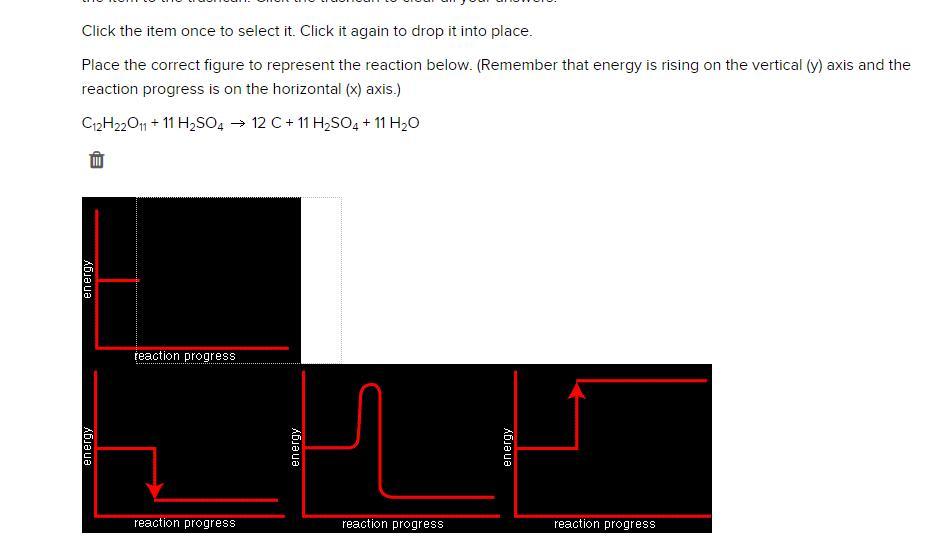
Place the correct figure to represent the reaction below. (Remember that energy is rising on the vertical (y) axis and the reaction progress is on the horizontal (x) axis.)
C12H22O11 + 11 H2SO4 12 C + 11 H2SO4 + 11 H2O
Answers
Answer:
It is the bottom left one
Explanation:
Just did it in OW
Related Questions
4 Hydrogen has three naturally occurring isotopes. H.1, H-2, and H-3 The atomie mass of Hydrogen is 1.097 Which isotope is most abundant in nature? Explain
Answers
Answer:
The number following the name of the element is the number of subatomic particles inside the nucleus of the atom. This means that it is the mass number of the isotope. The average atomic mass of the element is the sum of the products of the percentage abundance and mass number of the naturally occurring isotopes.
Since, the average atomic mass of the hydrogen is nearest to 1 then, the most abundant isotope should be hydrogen-1.
HELP PLEASE
What does the 2 mean in the formula of calcium nitrate? What does the 3
mean?
Answers
Answer:
Calcium nitrate is an inorganic compound with the chemical formula Ca(NO3)2. ... It is a nitrate salt of Calcium which contains calcium and nitrogen and oxygen. Calcium nitrate is a white or whitish-grey coloured granular solid which absorbs moisture from the air and is usually found as a tetrahydrate compound Ca(NO3)2.
Explanation:
Call each of your flowers as water employee increase rate of growth
Answers
Which is an example of a medicine that is made up of synthetic materials?
Fish Oil
Aspirin
Aloe Vera
Fresh water
Answers
Answer: Aspirin
Explanation:
What are the charges on ions of Group 1A, Group 3A (aluminum), and Group 5A?
Answers
Answer:
Group 1A: 1+
Group 3A: 3+
Group 5A: 3+ or 5+
Explanation:
An ideal gas occupies a volume V at an absolute temperature T. If the volume is halved and the pressure kept constant, what will happen to it's temperature?
a. It will halve to T/2.
b. It will increase to 3T.
c. It will increase to 2T.
d. It will remain the same.
Answers
Answer:
It will be halve of T
Explanation:
V1 = V
T1 = T
V2 = ½V
T2 = x
V1/T1 = V2/T2
V/T = ½V/x
Vx = ½VT
2Vx = VT
2x = T
x = ½T
I’ll really appreciate it if you help me on this one .
Answers
Answer:
Beautiful, scary, a different experience, full of imaginations.
Which conversion factor below would you use to complete this conversion?
How many liters of bromine gas would 12.3 moles occupy at Standard Temperature and Pressure?
a.) 1 mole/22.4 L
b.) 22.4 mol/1 liter
c.) 22.4 L/1 mole
d.) 1 liter/22.4 mol
Answers
Answer:
c.) 22.4 L/1 mole
Explanation:
Using the general gas law equation as follows:
PV = nRT
Where;
P = pressure (atm)
V = volume (L)
n = number of moles (mol)
R = gas law constant (0.0821 Latm/molK)
T = temperature (K)
The standard values for a gas are as follows:
Standard pressure of a gas = 1 atm
Standard temperature of a gas = 273K
According to the information in the question, n = 12.3 moles
Using PV = nRT
V = nRT/P
V = 12.3 × 0.0821 × 273/1
V = 275.68 L
Based on this value of volume in liters for 12.3moles of Br gas, the volume per mole at standard temperature and pressure will be 275.68/12.3 = 22.4 Litres per mole. Hence, the answer is 22.4 L/1 mole.
determine the volume in liters of metal sample weighting 352.2g and has a density of 7.10gmL.
Answers
Answer:
V = 0.0496 L
Explanation:
Given that,
The mass of a sample, m = 352.2 g
The density pf sample, d = 7.10 g/mL
We need to find the volume of the sample. We know that the density of an object is given by :
[tex]d=\dfrac{m}{V}\\\\V=\dfrac{m}{d}\\\\V=\dfrac{352.2}{7.1}\\\\V=49.6\ mL[/tex]
or
V = 0.0496 L
So, the volume of the sample is 0.0496 L.
Can someone pls help me with this its due in 20 minutes.
Answers
Answer:
15. The velocity of a sound wave is affected by two properties of matter: the elastic properties and density.
16. The greater the density of a medium, the slower the speed of sound. This observation is analogous to the fact that the frequency of a simple harmonic motion is inversely proportional to m, the mass of the oscillating object.
17. Temp °C 0 m/s2 m/s
50 360.51 361.62
18. Molecules at higher temperatures have more energy, thus they can vibrate faster. Since the molecules vibrate faster, sound waves can travel more quickly. The speed of sound in room temperature air is 346 meters per second. ... The speed of sound is also affected by other factors such as humidity and air pressure.
Air with less than 19.5% oxygen cannot support human life. How many moles of oxygen would the caisson if the percent of oxygen in the air dropped to 19.4%.
Answers
3. Identify the 4 primary pigments found in plant leaves (name and color)
Answers
Anthocyanins: red
Carotene: orange.
Sorry I couldn’t find the 4th one
balance
Na2O+H2O → NaOH
Answers
Na2O+H2O=2NaOH
Step by step exp.
Given:
Equation Na2O+H2O=NaOH
To find: Balance the equation
Solution:
Taking LHS of the equation
LHS=Na2O+H2O
There is 2 sodium, 2 oxygen,& 2 hydrogen
To balance the equation we have equal number of atom so we multply 2 to the RHS=2NaOH
There fore the equation form is
Na2O+H2O=2NaOH
What do u mean by electronic configuration? What are basic requirements while writing electronic configuration of an element (atom) ?
Answers
The basic requirement while writing the electronic configuration of an element (atom) is its atomic number.
Explanation:
The electronic configuration of an element is the distribution of its electrons in different energy levels around the atomic nucleus.It only requires an atomic number of an element to write its electronic configuration.The atomic number is equal to the number of protons in an atom and that is equal to the number of electrons.And the filling of electrons is done according to the Aufbau principle, the electrons will be first filled in the atomic orbitals with lower orbital energy before filling the atomic orbital with higher energy.The sequence of the atomic orbitals from lower energy to higher energy is:[tex]1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d...[/tex]
For example:
The atomic number of gallium is 31, its electronic configuration will be:
[tex][Ga]=1s^2 2s^2 2p^63s^2 3p^6 4s^2 3d^{10} 4p^1[/tex]
Learn more about an electronic configuration of an atom here:
brainly.com/question/11628377
brainly.com/question/18444398
This is 10th grade Chemistry
Answers
If the volume of an ideal gas is 22.5 L when it is at a temperature of 365 K, what will the volume be when the gas cools to 338K? Be sure to show your work and proper units.
Answers
Answer:
20.8L = Final volume of the gas
Explanation:
Based on Charles's law, the volume of a gas is directly proportional to the temperature of the gas under pressure constant. The equation is:
[tex]\frac{V_1}{T_1} =\frac{V_2}{T_2}[/tex]
where V is volume and T absolute temperature of 1, initial state and 2, final state.
If initial volume is 22.5L, initial T = 365K and final temperature 338K:
22.5L / 365K = V₂ / 338K
20.8L = Final volume of the gasRead the description of the centrioles. What is their function?
Answers
Answer:
Typically found in eukaryotic cells, centrioles are cylindrical (tube-like) structures/organelles composed of microtubules. In the cell, centrioles aid in cell division by facilitating the separation of chromosomes. For this reason, they are located near the nucleus.
Our solar system formed from a
) a. huge disk of dust and gases
b.milky way and stars
c. sun and a moon
Answers
If Steve throws a football 57 meters in 3 seconds, what is the average speed of the football?
Answers
Answer:
19 m/s
Explanation:
57/3
the symbol between two reactants in a chemical equation?
Answers
Hope this helps!!
Select the chemical formula that contains 10 atoms of hydrogen.
2C4H10
2CH4
2C2H5
2C2H6
Answers
Name the type of light interaction feeling hotter in a black shirt than a white shirt
1. Reflected
2. Absorbed
3. Transmittied
Answers
If 30.0 grams of calcium metal react with 18.0 grams of oxygen gas, your calculations show that ________ grams of CaO could be produced from the 30.0 grams of calcium and __________ grams of CaO could be produced from the 18.0 grams of oxygen. The theoretical yield of calcium oxide from this reaction is ________.
Answers
Answer:
42g if CaO can be produced from 30g and 31.5g of CaO will be produced from 18g of O
Explanation:
40g of CA =56g
30g =×
cross multiple
=56*30/40
=42g
32g of O =56g
18g =×
cross multiple
=56*18/32
=31.5g
theoretical yield
42-31.5= 10.5
just guessing
HELP ME!!!
Project: Modeling potential and kinetic energy
Assignment Summary
For this assignment, you will develop a model that shows a roller coaster cart in four different positions on a track. You will then use this model to discuss the changes in potential and kinetic energy of the cart as it moves along the track.
Background Information
The two most common forms of energy are potential energy and kinetic energy. Potential energy is the stored energy an object has due to its position. Kinetic energy is the energy an object has due to its motion. An object’s kinetic energy changes with its motion, while its potential energy changes with its position, but the total energy stays the same. If potential energy increases, then kinetic energy decreases. If potential energy decreases, then kinetic energy increases.
Potential energy related to the height of an object is called gravitational potential energy. Gravitational potential energy is directly related to an object’s mass, the acceleration due to gravity, and an object’s height.
Materials
One poster board per student Drawing utensils
Assignment Instructions
Step 1: Prepare for the project.
a) Read the entire Student Guide before you begin this project.
b) If anything is unclear, be sure to ask your teacher for assistance before you begin.
c) Gather the materials you will need to complete this project.
Step 2: Create your poster.
a) On the poster board, draw a roller coaster track that starts with one large hill, then is followed by a valley and another, smaller hill.
b) Draw a cart in four positions on the track as outlined below.
i. Draw the first cart at the top of the first hill. Label it A.
ii. Draw the second cart going down the first hill into the valley. Label it B.
iii. Draw the third cart at the bottom of the valley. Assume that the height of the cart in this position is zero. Label it C.
iv. Draw the last cart at the top of the second, smaller hill. Label it D.
c) Make sure that your name is on the poster. Step 3: Type one to two paragraphs that describe the energy of the cart.
a) Type one to two paragraphs describing the changes in potential and kinetic energy of the cart. Be sure to discuss how the potential and kinetic energy of the cart changes at each of the four positions along the track, and explain why these changes occur.
b) Make sure your name is on the document.
c) Later, you will submit this document through the virtual classroom.
Step 4: Evaluate your project using this checklist.
If you can check each criterion below, you are ready to submit your project.
Did you draw a model of a roller coaster track with one large hill, a valley, and a smaller hill?
Did you draw a cart on the track in the four required positions A–D? Did you label the cart at each of the four positions?
Did you type a paragraph describing the changes in potential and kinetic energy of the cart at each of the four positions on the roller coaster track? Did you explain why the changes in potential and kinetic energy occur?
Step 5: Revise and submit your project.
a) If you were unable to check off all of the requirements on the checklist, go back and make sure that your project is complete.
b) When you have completed your project, submit your poster to your teacher for grading. Be sure that your name is on it.
c) Submit the typewritten document through the virtual classroom. Be sure that your name is on it.
Step 6: Clean up your work space.
a) Clean up your work space. Return any reusable materials to your teacher and throw away any trash.
b) Congratulations! You have completed your project.
Answers
Answer:
Assignment Summary
For this assignment, you will develop a model that shows a roller coaster cart in four different positions on a track. You will then use this model to discuss the changes in potential and kinetic energy of the cart as it moves along the track.
Background Information
The two most common forms of energy are potential energy and kinetic energy. Potential energy is the stored energy an object has due to its position. Kinetic energy is the energy an object has due to its motion. An object’s kinetic energy changes with its motion, while its potential energy changes with its position, but the total energy stays the same. If potential energy increases, then kinetic energy decreases. If potential energy decreases, then kinetic energy increases.
Potential energy related to the height of an object is called gravitational potential energy. Gravitational potential energy is directly related to an object’s mass, the acceleration due to gravity, and an object’s height.
Materials
One poster board per student Drawing utensils
Assignment Instructions
Step 1: Prepare for the project.
a) Read the entire Student Guide before you begin this project.
b) If anything is unclear, be sure to ask your teacher for assistance before you begin.
c) Gather the materials you will need to complete this project.
Step 2: Create your poster.
a) On the poster board, draw a roller coaster track that starts with one large hill, then is followed by a valley and another, smaller hill.
b) Draw a cart in four positions on the track as outlined below.
i. Draw the first cart at the top of the first hill. Label it A.
ii. Draw the second cart going down the first hill into the valley. Label it B.
iii. Draw the third cart at the bottom of the valley. Assume that the height of the cart in this position is zero. Label it C.
iv. Draw the last cart at the top of the second, smaller hill. Label it D.
c) Make sure that your name is on the poster. Step 3: Type one to two paragraphs that describe the energy of the cart.
a) Type one to two paragraphs describing the changes in potential and kinetic energy of the cart. Be sure to discuss how the potential and kinetic energy of the cart changes at each of the four positions along the track, and explain why these changes occur.
b) Make sure your name is on the document.
c) Later, you will submit this document through the virtual classroom.
Step 4: Evaluate your project using this checklist.
If you can check each criterion below, you are ready to submit your project.
Did you draw a model of a roller coaster track with one large hill, a valley, and a smaller hill?
Did you draw a cart on the track in the four required positions A–D? Did you label the cart at each of the four positions?
Did you type a paragraph describing the changes in potential and kinetic energy of the cart at each of the four positions on the roller coaster track? Did you explain why the changes in potential and kinetic energy occur?
Step 5: Revise and submit your project.
a) If you were unable to check off all of the requirements on the checklist, go back and make sure that your project is complete.
b) When you have completed your project, submit your poster to your teacher for grading. Be sure that your name is on it.
c) Submit the typewritten document through the virtual classroom. Be sure that your name is on it.
Step 6: Clean up your work space.
a) Clean up your work space. Return any reusable materials to your teacher and throw away any trash.
b) Congratulations! You have completed your project.
Explanation:
list 5 things which we can make using each of the following materials
Answers
Answer:
Glass
1.microscopes
2.mirrors
3.glass shelves
4.wrist watch glass
5.magnifying glass
Metal
1.keys
2.automobile body parts
3.coins
4.window frames
5.screws and nails
Plastic
1.bottles
2.boxes
3.balls
4.carry bags
5.buckets and mugs
Wood
1.chairs
2.tables
3.shoe stand
4.dressing table
5.shelves
as insoluble ...... can be separated from a liquid by filtration on centrifugation
Answers
Answer:
by centrifugation
Explanation:
in such cases filtration cannot be used for separation
Answer the multiple choice question below
Which physical property allows liquid-liquid mixtures to be separated using filtration?
I
A. Melting point
В.
Density
C.
Flammability
Evaporation
Answers
Answer:
flammability evaporation
Pleade help me! (If you guys unfortunately I am going to report you) Thank you
Answers
Answer:
answer 3
Explanation:
For the reaction 2h2+o2>h20, how many grams of water are produced from 70 grams of hydrogen
Answers
630g
Explanation:
firstly the equation has to be balanced. 2H2+o2>2H2o.
finding the mole of hydrogen?
mole = 70/2 which is equal to 35 mole of hydrogen.
35 mole of hydrogen is equal to the mole of water.
finding the mass of water=?
35=mass/18
mass= 35*18=630g of water
True or False: The reactants and the products of a chemical equation always have the same number of atoms.
Answers
Answer: True
Explanation: It always has the same number because the both have or are using the same product.