is photosynthesis part of chemical change?If yes can I have photosynthesis in chemical equation as chemical symbols
Answers
Answer:
yes it is a chemical change
i have made it in above picture
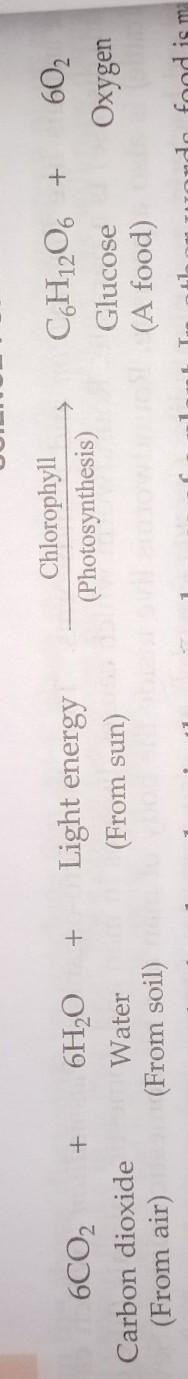
Related Questions
Measurements show that unknown compound X has the following composition:
element mass %
carbon 74.8%
hydrogen 25.1%
Write the empirical chemical formula of X. please help
Answers
the empirical chemical formula of X is CH4
Explanation:Step 1: Imagine you have a sample of compound weighing exactly . Multiply the mass of this sample by the mass percents to find the mass of each element in the sample
Step 2: Divide the mass of each element by the element's molar mass to find the moles of each element in the sample. Remember to round your answers to the correct number of significant digits.
Step 3: Divide the moles of each element by the the smallest number of moles of any element to find the mole ratio of elements in the sample.
Step 4: Multiply the mole ratio by the smallest whole number that changes it into a whole number ratio to find the atom ratio of elements in the sample.
Note that the result of each multiplication must equal a whole number only within measurement uncertainty.
The measurement uncertainty in this calculation comes from the measurement uncertainty of the mass percents given in the question. The mass percents each have significant digits. That means each mass percent has some measurement uncertainty in the third significant digit, and only the first two significant digits can be considered completely reliable.
In Step 1 you multiplied each mass percent by something with zero uncertainty (the exactly you assumed your sample weighed), in Step 2 you divided by a measurement with more than significant digits (the molar mass of the elements), and in Step 3 you divided by a measurement with the same number of significant digits (the least number of moles of any element in the compound). None of these steps added to the measurement uncertainty of your calculation.
Therefore, the measurement uncertainty in the final result of all your calculations is determined by the measurement uncertainty in the original mass percents and will be in the third significant digit. That means the result of each of the final multiplication steps must equal a whole number only to within the first two significant digits.
The whole numbers in the last column of the table are and .
which of the following has more particles
6.02x10(small numbers)23 molecules CO2
9 moles PF2
10 mole NaCI
18 g H2O
Answers
Answer:
A mole is Avogadro's number of items: 6.022 × 1023.
Explanation:
PLS HELP URGENT
Electron dot diagrams
Use your periodic table to write the electron dot diagrams for the following atoms.
1. Calcium (Ca)
2. Polonium (Po)
3. Moscovium (Mc)
4. Boron (B)
5. Fluorine (F)
Answers
A reaction rate is the change in of a reactant or product with
Answers
Answer:
the rate of reaction is the speed at which a chemical reaction takes place
What is an equilibrium constant?
A. A constant relating the effects of activation energy and temperature on the reaction rate
B. The value of the rate of the forward and reverse reactions at equilibrium
C. The amount of energy required for the collision of two molecules to result in a reaction
D. The ratio of equilibrium concentrations of products to concentrations of reactants
Answers
Answer:
Explanation:well first you gotta animalize in your brain what you think would happen in this case D would explain what an equilibrium constant is
Answer: D
Explanation:
The ratio of equilibrium concentrations of products to concentrations of reactants
identify NaOH (aq) + HNO3(aq) → NaNO3(aq) + H2O(1)
Answers
Answer:
this is called acid base reaction
Which om sled below is amplo dl an abelis laeler in an evünment?
ban
B. droganty
C. ar temperatura
D. cation
Answers
Answer:
Explanation:
i got D
Hope this helped, please make me brainliest if it was useful! Have a good day/night
Which condition would cause an increase in the salinity of ocean water?
thawing of glaciers
high rates of evaporation
cold weather
a river flowing into the sea
Answers
Answer:
high rates of evaporation
How many grams of Sulfur are there in 2.05 x 10^25 molecules of Calcium Sulfate?
Answers
Answer:
1090 grams Sulfur (3 sig-figs)
Explanation:
Given 2.05 x 10²⁵ molecules CaSO₄ => 2.05 x 10²⁵ Sulfur atoms (subscript = 1 mole).
Converting 2.05 x 10²⁵ sulfur atoms to moles, divide by Avogadro's Number
(= 6.023 x 10²³ sulfur atoms / mole sulfur) => 2.05 x 10²⁵ sulfur atoms/6.023 x 10²³ sulfur atoms/mole sulfur atoms = 34.036 moles sulfur atoms.
Converting 34.036 moles sulfur to grams sulfur multiply by formula wt. of sulfur (=32g/mole S)
=> 34.036moles S x 32g/mole S = 1089.158 grams S ≅ 1090 g S (3 sig-figs)
i really need help, this is important to me, if not i get my harry styles tickets taken away and if that happens my life is over lol, i’m not saying it for sympathy i just REALLY need help
Answers
Explanation:
First Question
Group 18 elements. This is because the noble elements do not conduct electricity.
Second Question
Chewing in the mouth breaks food into small pieces
Third Question
How many atoms of each element are present in the molecule?
7 carbon atoms, 14 Hydrogen atoms and 1 oxygen atom
A tank 20.0cm by 50.0cm by 30.0cm will hold how many liters of water?
Answers
Answer:
30,000 cm^3
Explanation:
volume for rectangular prism is v=l×w×h
plug in known values and solve.
v=(30.0cm)(20.0cm)(50.0cm)=30,000cm^3
How many moles of carbon dioxide are produced when 5.12 moles of glucose undergoes a combustion reaction?
Answers
Answer: 30.72 moles of [tex]CO_2[/tex] will be produced from 5.12 moles of glucose.
Explanation:
The balanced chemical equation for combustion of glucose is:
[tex]C_6H_{12}O_6+6O_2\rightarrow 6CO_2+6H_2O[/tex]
According to stoichiometry :
1 moles of glucose produce = 6 moles of [tex]CO_2[/tex]
Thus 5.12 moles of glucose produce =[tex]\frac{6}{1}\times 5.12=30.72moles[/tex] of [tex]CO_2[/tex]
Thus 30.72 moles of [tex]CO_2[/tex] will be produced from 5.12 moles of glucose.
When 10 grams of water completely evaporate into water vapor, the volume of the water vapor is greater than the volume of the original liquid water, but the mass remains the same. Why does the mass stay the same? Which answer is correct?
A
The temperature of the liquid water and water vapor is the same.
The temperature of the liquid water and water vapor is the same.
B
The density of the liquid water is the same as the density of the water vapor.
The density of the liquid water is the same as the density of the water vapor.
C
The number of atoms in the water vapor is equal to the number of atoms in the liquid water.
The number of atoms in the water vapor is equal to the number of atoms in the liquid water.
D
The distance between the water molecules is the same in the liquid and the vapor.
The distance between the water molecules is the same in the liquid and the vapor.
Answers
Answer: I think the answer is B or C, its kind of hard.
As the number of atoms in the water vapor is equal to the number of atoms in the liquid water. Therefore, the mass of the water remains the same.
What is evaporation?Evaporation is a process that usually happens on the surface of water or liquid and it involves the conversion of the liquid phase into the gaseous phase. This process involves a change in the state of matter of water or liquids.
When the water is completely evaporated into water vapor the density of the water vapor is less than the density of the liquid water as the vapors occupy a large volume in comparison to liquid water.
But the number of atoms in the water vapor is the same as the number of atoms in the liquid water. The mass of the water is due to the mass of the atoms.
Therefore, the mass of the water remains the same during the phase transition from liquid water to water vapors. Therefore, option (C) is correct.
Learn more about evaporation, here:
https://brainly.com/question/5019199
#SPJ2
Can you bond 2 silicon atoms (Si) together to form a complete (real) molecule? Explain your reasoning
Answers
Answer: no
Explanation: because silicon cannot share over 3 bonds
Chlorine atom
10. What does the following equation represent; 2KMnO4 -> K2MnO4 + MnO2(s)
+ O2(g) ?*
A. Oxygen turned in to carbon dioxide
B. Sulphate and lithium boiled
C. Decomposition of potassium permanganate(Heating)
D. None of the above
Answers
Answer:
C. Decomposition of potassium permanganate(Heating)
Explanation:
The equation of the reaction is given as;
2KMnO4 -> K2MnO4 + MnO2(s) + O2(g)
Reactant = 2KMnO4
Products = K2MnO4 + MnO2(s) + O2(g)
A. Oxygen turned in to carbon dioxide
Incorrect option - Oxygen is not the reactant
B. Sulphate and lithium boiled
Incorrect option - Sulphate and lithium are not part of this reaction
C. Decomposition of potassium permanganate(Heating)
Correct option - potassium permanganate decomposed to form K2MnO4 + MnO2(s) + O2(g)
D. None of the above
Incorrect option
word equation for HgO → Hg +O2
Answers
Please help me on this.
Answers
How many significant digits are in this number?
742800
1.6
2.5
3.4
4.3
Answers
what is a salient factor?
Answers
Answer:
technology is a silent factor
Explanation:
learned about it in computer class.
High-speed winds transfer more energy to the ocean’s surface, creating bigger waves.
Question 10 options:
True
False
Answers
Answer:
I think that would be true. The faster the wind, the longer it blows, or the farther it can blow uninterrupted, the bigger the waves. Therefore, a wave's size depends on wind speed, wind duration, and the area over which the wind is blowing
Answer: True.
Explanation: Yes, waves get energy from the wind through friction, so you see those waves get bigger each time. The longer the winds blows, the more friction, and energy it's getting, meaning better, and deadly waves. The wind can sometimes cause tsunamis if the its blowing too hard.
2. Hydrocarbons consist of carbon and hydrogen. 5.7 g of a hydrocarbon contains 4.8 g of carbon.
If the relative molecular mass of the hydrocarbon is 114, determine its molecular formula.
Answers
Answer:
The molecular formula of the hydrocarbon compound is C₈H₁₈
Explanation:
The type of chemical substance in the question = Hydrocarbon
The mass of the hydrocarbon = 5.7 g
The mass of carbon in the given hydrocarbon (sample) = 4.8 g
The relative molecular mas of the hydrocarbon = 114 g
Therefore, the mass of hydrogen in the sample, H = 5.7 g - 4.8 g = 0.9 g
The ratio of the mass of carbon in the sample = 4.8/5.7
The ratio of the mass of hydrogen in the sample = 0.9/5.7
By the law of constant composition, we have;
The mass of carbon in a mole of the hydrocarbon = (4.8/5.7) × 114 g = 96 g
The molar mass of carbon, C = 12 g/mol
The number of moles of carbon in a mole of the hydrocarbon, 'n₁', is given as follows;
n₁ = 96 g/12 g = 8 moles
The mass of hydrogen in a mole of the hydrocarbon = (0.9/5.7) × 114 g = 18 g
The molar mass of hydrogen, H ≈ 1 g/mol
The number of moles of hydrogen in a mole of the hydrocarbon, 'n₂', is given as follows;
n₂ = 48 g/1 g = 18 moles
Therefore, in each each molecule of the compound, we have have 8 atoms of carbon, 'C', and 18 atoms of hydrogen, 'H'
Therefore, we get;
The molecular formula of the hydrocarbon compound is C₈H₁₈ which is the chemical formula for the hydrocarbon also known as octane
Which of the following best describes a solution?
a heterogenous compound
a homogenous compound
a homogenous mixture
O O a heterogenous mixture
Answers
Answer:
homogeneous mixture
Under each photo,
write an example of where you
might find water in that state of
matter.
Answers
Answer:
Explanation:
water vapor:in a cloud or when you breath out once it's cold.Ice: in your freezer or on a frozen lake.liquid water:at a stream
How many kilograms of water must be added to 6.07 grams of oxalic acid (H2C2O4) to make a 0.025 m solution?
Answers
M
a
V
a
=
M
b
V
b
M
a
= 6.77M - the initial molarity (concentration)
V
a
= 15.00 mL - the initial volume
M
b
= 1.50 M - the desired molarity (concentration)
V
b
= (15.00 + x mL) - the volume of the desired solution
(6.77 M) (15.00 mL) = (1.50 M)(15.00 mL + x )
101.55 M mL= 22.5 M mL + 1.50x M
101.55 M mL - 22.5 M mL = 1.50x M
79.05 M mL = 1.50 M
79.05 M mL / 1.50 M = x
52.7 mL = x
59.7 mL needs to be added to the original 15.00 mL solution in order to dilute it from 6.77 M to 1.50 M.
I hope this was helpful.
What are two methods used to help identify the species of insect from the eggs on
human remains? Describe the methods.
Answers
Answer:
The two different methods to help identify the species of insects from the eggs on human remains are by using a small paint brush dipped in water and the other way is by using forceps.Explanation:
mark as brainliestThe insects have been defined as the most successful arthropods. There are far more species in the class Insecta than in any other group of animals. The insects are the only invertebrates with wings.
What are insects?The insects play a very important role in the web of the life in every environment. Their jobs include pollinating flowering plants, since it is the source of food for insectivorous animals and it assist in the decomposition of plants and animals.
The two different methods which help to identify the species of the insects from the eggs on human remains are by using a small paint brush dipped in water and the other way is by using the forceps.
The study of insects and their life cycle are found to be important for the forensic entomologist. This is because it could set a timeline on when the crime is happened. The life cycle of a blowfly starts as an egg and then goes to be maggots.
To know more about insects, visit;
https://brainly.com/question/13277346
#SPJ2
Please help pretty please
Answers
Answer: Its is a field generated by any two objects touching each other.
OR
Answer: Its is a field generated by any object with any mass.
Explanation: I AM SOOOO SORRY if i get it wrong i pretty sure the first one is right i mean like My first ansewr i typed in......... :/ :/
100 grams of liquid iron is put in liquid water when is equilibrium reached
Answers
Answer:
when the water heats up i believe
Explanation:
b
Which statement is not
true?
A. The troposphere is responsible for nearly
all of Earth’s weather.
B. The exosphere reaches deep into space
and is the least dense layer.
C. The thermosphere typically breaks up
meteors before they hit Earth.
D. The stratosphere allows commercial
airlines to fly with less turbulence because
of fewer convection currents.
Answers
explanation: to make C correct, the sphere that breaks up meteors is the mesosphere.
The atmosphere of the earth is layered and each layer of the atmosphere has its own properties. The atmosphere of the earth is divided into four layers. The thermosphere typically breaks up meteors before they hit Earth is not correct. The correct option is C.
What is mesosphere?The four important layers of atmosphere are troposphere, stratosphere, thermosphere and mesosphere. The layer of the atmosphere which is found above the stratosphere is defined as the mesosphere. It is the coldest layer of the atmosphere.
A meteor is known as a celestial object which is made up of rocks and minerals which enters the atmosphere of the earth and burns out completely before reaching the surface of the earth. It is known as the shooting star or falling star.
The temperature of mesosphere drops with altitude. By 80 km it reaches 100 degree celsius. The meteors burn up in this layer not in thermosphere.
Thus the correct option is C.
To know more about earth layers, visit;
https://brainly.com/question/13497783
#SPJ3
word equation for Na2O+H2O → NaOH
Answers
Answer:
Sodium oxide + Water - - - > Sodium Hydroxide.
The Moon is always half lit and half dark.
True or
False
(i know this isn’t chemistry theres no science one.)
Answers
Answer:
True
Explanation:
Just like the Earth, half of the Moon is lit by the Sun while the other half is in darkness.
Answer:
Pretty sure it's true.
On Earth, we only see part of the moon that is illuminated or not, since it revolves around the Earth. However, the sun is stationary and doesn't move, meaning that one side of the moon is always lit by the sun as it revolves around the Earth.