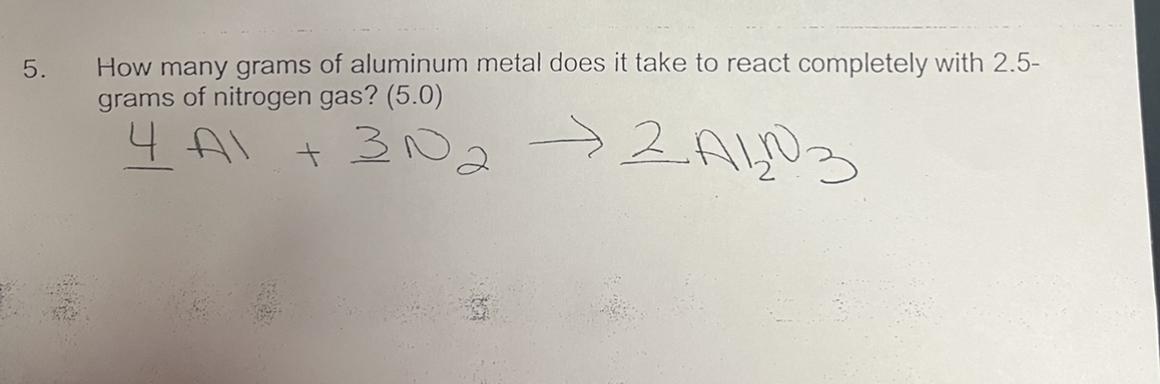Answers
In this stoichiometry question, we have the following reaction:
4 Al + 3 N2 -> 2 Al2N3
We have:
2.5 grams of N2
We want to know how much Al we need in order to react with 2.5 grams of N2, and in order to do that, we need to find out how many moles of N2 we have in 2.5 grams, we will be using its molar mass, 28g/mol to do it:
28g = 1 mol
2.5g = x moles
28x = 2.5
x = 2.5/28
x = 0.089 moles of N2 in 2.5 grams
Now according to the molar ratio, we have 4 moles of Al for every 3 moles of N2, therefore, if we have 0.089 moles of N2:
4 Al = 3 N2
x Al = 0.089 N2
x = 0.119 moles of Al
Now we can find the mass needed, using the number of moles, 0.119 moles, and also the molar mass, 27g/mol:
27g = 1 mol
x grams = 0.119 moles of Al
x = 3.2 grams of Al is required
Answer is 3.2 grams
Related Questions
Force and distance are used to calculate work. Work is measured in which unit?
Answers
Answer: Joules (J)
Explanation:
Answer:
Joules or Newton metre
Explanation:
Work = Force × distance
Force = kgm/s² (Newton)
×
Distance = m
= kgm²/s² (Newton metre or Joules) (Nm or J)
5 F2 (g) + 2 NH3 (g) → N2F4 (g) + 6 HF (g)If you reacted 7.78 moles of F2 how many moles of HF would be produced?
Answers
5 F2 (g) + 2 NH3 (g) → N2F4 (g) + 6 HF (g)
2. Determine mole proportions , then solveFrom the above balanced reaction, we can see that :
5 moles of F2 reacts to form : 6 moles of HF ,
so, 7.78moles F2 will give :x moles of HF
Therefore,x mole HF =( 6 moles HF * 7.78 mole F2)/ 5 mole F2
= 6 * 7.78/5
=9.336
≈9.34moles HF
• This means that , if you reacted 7.78 moles of F2 , ,9.34 moles of HF would be produced,.
Calculate how many grams of MgCl2 are needed to produce 12.15 grams
Answers
Answer
47.60 g
Explanation
Given that:
Molar mass of MgCl₂ = 95.20 g/mol
Mass of Mg produced = 12.15 grams
Equation: MgCl₂ → Mg + Cl₂
What to find:
To calculate the grams of MgCl₂ needed to produce 12.15 grams of Mg.
Step-by-step solution:
Step 1: Convert the mass of Mg produced to moles.
Using the molar mass of Mg (24.305 g/mol) and the mole formula, the moles of Mg produced is
[tex]Moles=\frac{mass}{molar\text{ }mass}=\frac{12.15\text{ }g}{24.305\text{ }g\text{/}mol}=0.49989714\text{ }mol[/tex]Step 2: Determine the moles of MgCl₂ needed to produce 12.15 g of Mg.
According to the given equation; 1 mol of MgCl₂ produced 1 mol of Mg
So, x mol of MgCl₂ will produce 0.49989714 mol of Mg
That is:
[tex]\begin{gathered} 1mol\text{ }MgCl₂=1mol\text{ }Mg \\ \\ x=0.49989714mol\text{ }Mg \\ \\ x=\frac{0.49989714mol\text{ }Mg}{1mol\text{ }Mg}\times1mol\text{ }MgCl₂ \\ \\ x=0.49989714mol\text{ }MgCl₂ \end{gathered}[/tex]Step 3: Convert the moles of MgCl₂ needed to produce 12.15 g of Mg to grams.
Using the molar mass of MgCl₂ = 95.20 g/mol, therefore the mass in grams of MgCl₂ needed to produce 12.15 g of Mg will be:
[tex]\begin{gathered} Mass\text{ }of\text{ }MgCl_2\text{ }needed=Molar\text{ }mass\times Moles \\ \\ Mass\text{ }of\text{ }MgCl_2\text{ }needed=95.20\text{ }g\text{/}mol\times0.49989714\text{ }mol \\ \\ Mass\text{ }of\text{ }MgCl_2\text{ }needed=47.60\text{ }g \end{gathered}[/tex]Hence, the grams of MgCl₂ needed to produce 12.15 grams of Mg is 47.60 g.
A gas sample in a closed, expandable container of initial volume 5.00 L was allowed to warm from 25 °C to 35°C. What was its new volume?
Answers
Taking into account the Charles' Law, a sample of gas in a closed expandable container with an initial volume of 5.00 L was allowed to warm from 25°C to 35°C to a volume of 5.168 L.
Charles' LawCharles' Law consists of the relationship that exists between the volume and the temperature of a certain amount of ideal gas, which is maintained at a constant pressure.
This law says that for a given sum of gas at a constant pressure, as the temperature increases, the volume of the gas increases and as the temperature decreases, the volume of the gas decreases because the temperature is directly related to the energy of motion of the gas molecules.
In summary, Charles's law is a law that says that when the amount of gas and the pressure remain constant, the ratio between the volume and the temperature will always have the same value:
V÷T=k
Considering an initial state 1 and a final state 2, it is fulfilled:
V₁÷ T₁= V₂÷ T₂
New volume in this caseIn this case, you know:
V₁= 5 LT₁= 25 °C= 298 KV₂= ?T₂: 35 °C= 308 KReplacing in Charles' law:
5 L÷ 298 K= V₂÷ 308 K
Solving:
(5 L÷ 298 K)× 308 K= V₂
5.168 L= V₂
Finally, the new volume is 5.168 L.
Learn more about Charles' law:
brainly.com/question/4147359
#SPJ1
2 NaOH + H2SO4 ------> 2 H2O + Na2SO4What is the mole ratio of H2O to H2SO4?
Answers
Answer
The mole ratio of H2O to H2SO4 is 2:1
Explanation
Given chemical equation:
[tex]2\text{NaOH }+H_2SO_4\rightarrow2H_2O+Na_2SO_4[/tex]From the balanced chemical equation above, 2 moles of NaOH react with sulfuric acid, 1 mole of H2SO4, to produce 2 moles of water and 1 mole sodium sulfate.
So, from the chemical equation, you have a 2:1 mole ratio between H2O and H2SO4
Calculate the molarity of a solution that contains 0.75 mol of KCI in 0.250L of solution
Answers
The molarity of a solution that contains 0.75 mol of KCI in 0.250L of the solution is 3 M
The number of moles per liter of solution is termed molarity.
M=n/V(in L)
M is the molarity
n is the number of moles
V is the volume
Substituting the values, we get
M=0.75/0.25=3
The molarity is 3 M
Molarity is a concentration term and denotes the number of moles dissolved per liter.It can be defined as the amount of substance in a particular volume of solution.The unit is mol/LIt can be found by the given mass and volume of the solution.To learn more about molarity visit:
https://brainly.com/question/8732513
#SPJ9
A chemical formula of an unknown metal (M) combines with chlorine in the followingformula:MC15What is the charge of the metal
Answers
Answer:
The charge of the metal is +5.
Explanation:
As the chlorine atom has a valence of -1, and the metals have a positive valence, we have to look for the charge that makes the molecule neutral.
In this case, there are 5 Cl, so the total negative charge of Cl is -5.
To make the molecule neutral, the metal must have a positive charge of +5.
Which of the following are organic compounds? (a). CH3CH3 (b). NH3 (c). CH3SCH3 (d). CH3CH2OH (e). Mg(OH)2
Answers
CH3CH3
CH3SCH3
CH3CH2OH
Explanations;What are organic compounds?
Organic compounds are compounds that class of compounds that contains the carbon-hydrogen bond. This means that for a compound to be classified as an organic compound, they must contain the C-H bond in their compound
From the listed compouds, the compound that contains theC-H bonds are:
• CH3CH3
,• CH3SCH3
,• CH3CH2OH
These are therefore considered the required list of organic compounds from the given options
What is the Mass in grams of 1 formula unit or molecule of epsom salt MgSO4●7H2O?
Answers
MgSO4.7H2O
First of all, we need to calculate the molar mass of this molecule, using the periodic table.
Let's take each atomic mass of the elements that form this molecule from the periodic table.
Mg = 24.30 u
S = 32.06
O = 15.99 u
H = 1.007 u
Now, we have to do this:
Molar mass (g/mol) = 1 x 24.30 + 1 x 32.06 + 4 x 15.99 + 14 x 1.007 + 7 x 15.99
Molar mass = 246.3 g/mol
We have to be careful with this result because 246.3 g is the mass of 1 mole of MgSO4.7H2O and we want the mass of 1 formula unit or molecule.
Pay attention to this:
1 mole of MgSO4.7H2O = 246.3 g = 6.022 x 10^23 formula units or molecules of MgSO4.7H2O
(We want the mass of 1 molecule and we have just calculated the mass of 6.022x10^23 molecules)
----------------------------------------------------------------------------------------------------------------
For 1 formula unit or molecule:
246.3 g of MgSO4.7H2O ------------- 6.022 x 10^23 formula units or molec.
x ------------- 1 formula unit or molec.
[tex]x\text{ = }\frac{1formula\text{ unit x 246.3 g}}{6.022x10^{23}formula\text{ units}}=4.09x10^{-22}g[/tex]Answer: The mass of 1 formula unit or molecule of MgSO4.7H2O = 4.09x10^-22 g
What is the charge on a sulfur atom that contains 18e−e−?
Answers
Sulphur atom has 16 as its atomic number, which is also the same number of protons and the same number of electrons:
Z = 16
P = 16
e- = 16
But when we have a Sulphur ion, it is usually -2, and since we have 18 electrons in this case, the charge will be -2
2C2H2 + 5O2 —> 4CO2 + 2H2OIf 3.80 moles of oxygen wre used up in the reaction, how many moles of water are produced?
Answers
In order to solve this question, we can use the concept of molar ratio, which means the amount of reactant is needed in order to produce a certain amount of product. In our question we have:
2 C2H2 + 5 O2 -> 4 CO2 + 2 H2O
The molar ratio between O2 and H2O is 5:2 (we use only the numbers in front of the compound), so that means that for every 5 moles of O2 used, we will have 2 moles of H2O being produce, now we can use this information to calculate how many moles of H2O will be produced from 3.80 moles of O2
5 O2 = 2 H2O
3.80 O2 = x H2O
x = 1.52 moles of H2O are produced from 3.80 moles of O2
Describe the solution if 274 g of potassium iodide (KI) is dissolved in 200 g of H2O at 10oC.
Answers
Which is the limiting reactant in the following chemical reaction: NH3 + 5 O2 —> 4 NO + 6 H2O, if 4 grams of O2 is used and 2 grams of NH3 is used ? Show work
Answers
The limitant reactant of a reaction is the reactant that we have the least number of mols considering its coefficient on the stoichiometry.
The reaction in this case is:
[tex]NH_3+5O_2\to4NO+6H_2O[/tex]The reactants are the one on the left side, so the answer can't be NO or H₂O.
The first thing to do is to calculate the number of moles of NH₃ and O₂, but for this we need their molar masses:
[tex]M_{NH_3}=1\cdot M_n+3\cdot M_H=(1\cdot14.0067+3\cdot1.00794)g/mol=17.03052g/mol[/tex][tex]M_{O_2}=2\cdot M_O=2\cdot15.9994g/mol=31.9988g/mol[/tex]Now, using them, let's calculate the number of moles of each:
[tex]\begin{gathered} M_{NH_{3}}=\frac{m_{NH_3}}{n_{NH_{3}}} \\ n_{NH_3}=\frac{m_{NH_3}}{M_{NH_{3}}}=\frac{2g}{17.03052g/mol}=0.11743\ldots.mol \end{gathered}[/tex][tex]\begin{gathered} M_{O_{2}}=\frac{m_{O_2}}{n_{O_{2}}} \\ n_{O_2}=\frac{m_{O_2}}{M_{O_{2}}}=\frac{4g}{31.9988g/mol}=0.12500\ldots mol \end{gathered}[/tex]Now, we can't compair directly, we need to consider their coefficients.
To do this, we divide the number of moles by the coefficient:
[tex]\begin{gathered} NH_3\colon\frac{n_{NH_3}}{1}=\frac{0.11743\ldots mol}{1}=0.11743\ldots mol \\ O_2\colon\frac{n_{O_{2}}}{5}=\frac{0.12500\ldots mol}{5}=0.025000\ldots mol \end{gathered}[/tex]Now, we can compair.
Since the value we have got to O₂ is less than NH₃, the limiting reactant is O₂
How many moles in 2.24 L of a sample of gas.1 mol10 mole0.1 mole5 mol
Answers
Answer:
[tex]0.1\text{ mole}[/tex]Explanation:
Here, we want to get the number of moles in 2.24 L of a sample of gas
Mathematically,
At STP, 1 mole of a gas occupies a volume of 22.4 L
Thus x moles will occupy 2.24 L at STP
To get the value of x, we have it that:
[tex]\begin{gathered} x\times22.4\text{ = 1}\times2.24 \\ x\text{ = }\frac{2.24}{22.4} \\ \\ x\text{ = 0.1 mole} \end{gathered}[/tex]Calculate the number of molecules in 8.21 x 10^-2 moles in (NH4)2Cr2O7
Answers
Answer
4.94x10^22 molecules
explanation
to calculate the number of molecules in 8.21x10^-2 moles ca be determined by using Avogadros number
we know that
1 mol has 6.022x10^23 molecules
therefore,
8.21x10^-2 moles have:
(8.21x10^-2) x (6.022x10^23)
= 4.94x10^22 molecules
Which of these elements are often made in a lab?
transuranium
noble gas
alkali metal
halogen
Answers
Answer:
I am sure it's Transuranium.
Explanation:
Hope it's right
Which pair of formulae represents two alkanes?a) CH4 and C8H18b) C2H6 and C5H8c) C3H6 and C5H12d) C10H8 and C4H8
Answers
Answer: the best option to answer the question is letter A
Explanation:
The question requires us to choose the option that presents the formula of two alkanes.
Alkanes are hydrocarbons that are saturated and acyclic (i.e., the molecule of an alkane do not present double or triple bonds between carbons and it is a linear structure). The general formula of alkanes tells us that there are double plus 2 the amount of hydrogen atoms as there is carbon atoms, as it follows:
[tex]C_nH_{2n+2}[/tex]where n indicates the number of carbon atoms in the molecule.
For example: we can say that the compound C2H6 is an alkane because it contains 2 carbon atoms (n = 2) and 6 hydrogen atoms (2n +2= 6).
Considering the options given by the question, only option A presents the formula of two alkanes:
CH4: n = 1; 2n+2 = 2+2 = 4
C8H18: n = 8; 2n+2 = 16+2 = 18
Therefore, the best option to answer the question is letter A.
The theoretical value isa) true valueOb) experimental valueOc) observed valueO d) measured value
Answers
Answer:
[tex]A\text{ : True value}[/tex]Explanation:
Here, we want to get another name for the theoretical value
The theoretical value is simply the value that has been accepted as true
Thus, we can call the theoretical value the true value
The following reaction takes place in an acidic solution. MnO4–(aq) + Cl–(aq) --> Mn2+ + Cl2(g) (unbalanced).Do Each Step Individually**: Write the half reactions. Balance the equations for atoms (except O and H). Balance the equations for atoms O and H using H2O and H+. Balance the charge in the half reactions. Multiply each half reaction by the proper number to balance charges in the reaction. Add the equations and simplify to get a balanced equation.
Answers
Answer
2MnO₄⁻(aq) + 10Cl⁻(aq) + 16H⁺ → 2Mn²⁺(aq) + 5Cl₂(g) + 8H₂O
Explanation
What is given:
The following reaction takes place in an acidic solution:
MnO₄⁻(aq) + Cl⁻(aq) → Mn²⁺(aq) + Cl₂(g) (unbalanced).
What to find:
To identify the half-reactions and balance the redox reaction.
Step-by-step solution:
Step 1: Identify the half-reactions.
The oxidation half-reaction is: Cl⁻(aq) → Cl₂(g)
The reduction half-reaction is: MnO₄⁻(aq) → Mn²⁺(aq)
Step 2: Balance the equations for atoms (except O and H).
Oxidation: 2Cl⁻(aq) → Cl₂(g)
Reduction: MnO₄⁻(aq) → Mn²⁺(aq)
Step 3: Balance the equations for atoms O and H using H₂O and H⁺.
Oxidation: 2Cl⁻(aq) → Cl₂(g)
Reduction: MnO₄⁻(aq) + 8H⁺ → Mn²⁺(aq) + 4H₂O
Step 4: Balance the charge in the half-reactions.
Oxidation: 2Cl⁻(aq) → Cl₂(g) + 2e⁻
Reduction: MnO₄⁻(aq) + 8H⁺ + 5e⁻ → Mn²⁺(aq) + 4H₂O
Step 5: Multiply each half-reaction by the proper number to balance charges in the reaction.
Oxidation: 2Cl⁻(aq) → Cl₂(g) + 2e⁻ x 5
Reduction: MnO₄⁻(aq) + 8H⁺ + 5e⁻ → Mn²⁺(aq) + 4H₂O x 2
↓
Oxidation: 10Cl⁻(aq) → 5Cl₂(g) + 10e⁻
Reduction: 2MnO₄⁻(aq) + 16H⁺ + 10e⁻ → 2Mn²⁺(aq) + 8H₂O
Step 6: Add the equations and simplify to get a balanced equation.
2MnO₄⁻(aq) + 10Cl⁻(aq) + 16H⁺ + 10e⁻ → 2Mn²⁺(aq) + 5Cl₂(g) + 8H₂O + 10e⁻
Simplifying the equation, we have:
2MnO₄⁻(aq) + 10Cl⁻(aq) + 16H⁺ → 2Mn²⁺(aq) + 5Cl₂(g) + 8H₂O
The molecular mass of a compound with an empirical formula of SO2 is 256 g/mol. What is the molecular formula?
Answers
So,
The molecular mass of the empirical formula of the compound SO2, can be found multiplying the molar mass of each element by the number of atoms and then sum. Let me explain this here below:
[tex]\begin{gathered} S\colon\frac{32g}{\text{mol}}\cdot1 \\ O\colon\frac{16g}{\text{mol}}\cdot2 \\ mSO_2=\frac{64g}{mol} \end{gathered}[/tex]We're given that the molecular mass of the molecular formula is 256g/mol. So, we could find the ratio between the mass of both formulas:
[tex]\frac{256}{64}=4[/tex]This means that the mass of the molecular formula is 4 times greater than the mass of the empirical formula.
Thus, the molecular formula will be:
[tex]S_4O_8[/tex]write any three chemical properties of carbon
Answers
Answer
• Carbon has the atomic symbol C,
,• Carbon is a nonmetallic chemical element with 4 electrons in its valence shell, which allows it to form 4 bonds forming compounds like CH₄.
,• Reactivity: Carbon does not dissolve in or react with water or acids.
,• The atomic mass of carbon is 12.011 g. It contains 6 protons in its nucleus.
If the reaction in the given equation produced 0.364 moles of H2 gas at STP, how many liters would this amount of H2 occupy? [hint - the H2 gas is at STP! Is there a special conversion we can use?]
Equation: 6 HCl + 2 Al ---> 2 AlCl3 + 3 H2
Answers
In the given equation produced 0.364 moles of H₂ gas at STP, 22.4 liters volume would this amount of H₂ occupy.
What is moles?The mole, denoted by the sign "mol," is the volume of a system that has the same number of atoms in 0.012 kilograms of carbon-12 as there are elementary particles.
The equation becomes:
6 HCl + 2 Al → 2 AlCl₃ + 3 H₂
6 mol 2 mol 2 mol 3 mol
As we know,
one mole of a gas occupies 22.4 liters.
Here, in the above equation, 3 moles of H₂ gas is produced.
So, in STP condition 3 moles of H₂ gas occupy = 3 × 22.4 lit
= 67.2 lit
Thus, in the given equation produced 0.364 moles of H₂ gas at STP, 22.4 liters volume would this amount of H₂ occupy.
To know more about volume refer to:
https://brainly.com/question/25693692
#SPJ1
5. What is the amount of moles of nitrous oxide gas (N2O) used to sedate Mr. Muthbefore his wisdom teeth are pulled? The pressure is 2.5 atm, a temperature of 25.and volume of 1.67L?tbd
Answers
Step 1 - Understanding how mole relates to temperature, pressure and volume
These variables are related according to the ideal gas equation:
[tex]pV=nRT[/tex]We can use this equation to predict the behavior as well as the quantity of a gas, given that the remaining variables are given.
Step 2 - Using the equation to solve the equation
According to the exercise:
[tex]\begin{gathered} p=2.5atm \\ T=25°C\text{ \lparen298 K\rparen} \\ V=1.67L \\ n=? \end{gathered}[/tex]Setting the values in the equation:
[tex]2.5\times1.67=nR\times298[/tex]We need to make two observations now: the temperature must be in Kelvin in this equation for it to work. R stands for the universal gas constant, as has the value of 0.082 atm.L/mol.K.
Therefore:
[tex]\begin{gathered} 2.5\times1.67=n\times0.082\times300 \\ \\ n=\frac{4.17}{24.6}=0.169\approx0.17 \end{gathered}[/tex]Answer: the amount of moles used is 0.17 moles.
The question is in the photo (Sorry for being lazy lol )
Answers
• When Sodium Sulfate is mixed with Barium chloride, the following reaction occurs :
BaCl2 (aq) + Na2SO4 (aq) ------> BaSO4 ( s) + 2 NaCl( aq)• a precipitate is the solid that forms, the white solide will be BaSO4
,• So option 4 is correct. BaSo4.
When salt dissolves in water, it shows the property of
Answers
Answer: Solubility
Explanation:
The property by which substances dissolve in water is called solubility. Soluble substances (such as salt) dissolve in water.
Answer: Electical charge
Explanation:At the molecular level, salt dissolves in water due to electrical charges and due to the fact that both water and salt compounds are polar, with positive and negative charges on opposite sides in the molecule.
What would the products of a double-replacement reaction between NaF andMgS be? (Remember: In double-replacement reactions, the two cationsswitch places and the two anions switch places.)O A. NaF and MgsB. MgF and NashO C. F2S and MgNa2O D. MgF2 and Na2SSUBMIT
Answers
Step 1
The reaction between NaF and MgS:
2NaF + MgS => Na2S + MgF2
(Remember: In double-replacement reactions, the two cations switch places and the two anions switch places.)
Answer: D. MgF2 and Na2S
A person has a 1.43mol sample of carbon dioxide. If the pressure if the sample is 34.56kpa, and the volume is 440.99mL, what will the temperature of the sample be in Kelvin?
Answers
Answer:
[tex]1.28K\text{ }[/tex]
Explanation:
Here, we want to get the temperature of the gas
We can use the ideal gas equation here
Mathematically, we have this as:
[tex]PV\text{ = nRT}[/tex]P is the pressure which is given as 34.56 kPa
V is the volume which is given as 440.99 mL
R is the molar gas constant which is 0.08205 L atm/mol k
n is the number of moles which is given as 1.43 mol
We need to convert the pressure to the correct unit (considering the molar gas consnat value in atm)
1 kPa = 0.00986923 atm
34.56 kPa will be = 34.56 * 0.00986923 = 0.341 atm
We need to convert the volume to Liters by dividing by 1000
We have that as 440.99/1000 = 0.44099 L
Finally,we rewrite the equation in terms of temperature and substitute the values as follows:
[tex]\begin{gathered} T\text{ = }\frac{PV}{nR} \\ \\ T\text{ = }\frac{0.341\times0.44099}{1.43\times0.08205}\text{ =1.28 K } \end{gathered}[/tex]A tank contains 8.5 L of fluorine at 268K and 549mm Hg. The gas is then transferred to a 5.0L tank. What temperature is required to obtain a final pressure of 675 mm Hg?
Answers
The temperature required to obtain a final pressure of 675 mm Hg194 is .86 K.
An ideal gas is a theoretical gas composed of many randomly transferring factor particles that aren't difficult to interparticle interactions. the best gasoline idea is beneficial because it obeys the precise gas law, a simplified equation of country, and is amenable to evaluation under statistical mechanics.
Volume is a degree of occupied three-dimensional space. it's far more frequently quantified numerically the usage of SI-derived gadgets or by way of diverse imperial gadgets. The definition of length is interrelated with the extent.
An ideal gas is described as one for which both the extent of molecules and forces between the molecules are so small that they have got no effect at the behavior of the gas.
Using the ideal gas equation:-
Given;
P₁ = 549mm Hg = 0.722369 atm
V₁ = 8.5 L
T₁ = 268 K
P₂ = 675 mm Hg = 0.89 atm
V₂ = 5
T₂ = ?
P₁V₁/T₁ =P₂V₂/T₂
T₂ = P₂V₂T₁/P₁V₁
= 0.89 × 5 × 268 / 8.5 × 0.72
= 194 .86 K
Learn more about ideal gas here:-https://brainly.com/question/20348074
#SPJ1
Standard temperature andpressure (STP) is [?] K and[ ] átm.
Answers
Remember that standard temperature and pressure (STP) is when the pressure is 1 atm and the temperature is 0 °C (273 K).
The answer is 273 K and 1 atm.
A balloon is filled to a volume of 1.50 L with 2.50 moles of gas at 25 °C. With pressure and temperature held constant, what will be the volume of the balloon if 0.20 moles of gas are added?
Answers
The final volume of the balloon if 0.20 moles of gas are added will be 1.62 L.
What will be the volume of the balloon given that 0.20 moles of a gas is added to it?The volume of the balloon given that 0.20 moles of a gas is added to it is determined using the ideal gas equation as follows:
P₁V = nRT
Where;
P is pressureV is volumen is the number of moles of gasR is molar gas constant = 0.082 atm.L/K.molInitial volume, V₁ = 1.50 L
Initial mole of gas, n₁ = 2.50 mol
Initial temperature, T₁= 25°C or 298 K
Initial pressure, P₁ = RT₁n₁/V₁
P₁ = 0.082 * 298 * 2.50 /1.50
P₁ = 40.7 atm
With pressure and temperature held constant:
Final temperature, T₂ is equal to T₁ = 298 K
Final pressure, P₂ is equal to P₁ = 40.7 atm
Final moles of gas, n₂ = 0.20 + 2.5
n₂ = 2.70 mol
Final volume, V₂ = RT₂n₂/P₂
V₂ = 0.082 * 298 * 2.70 /40.7
V₂ = 1.62 L
Learn more about gas volume at: https://brainly.com/question/27100414
#SPJ1