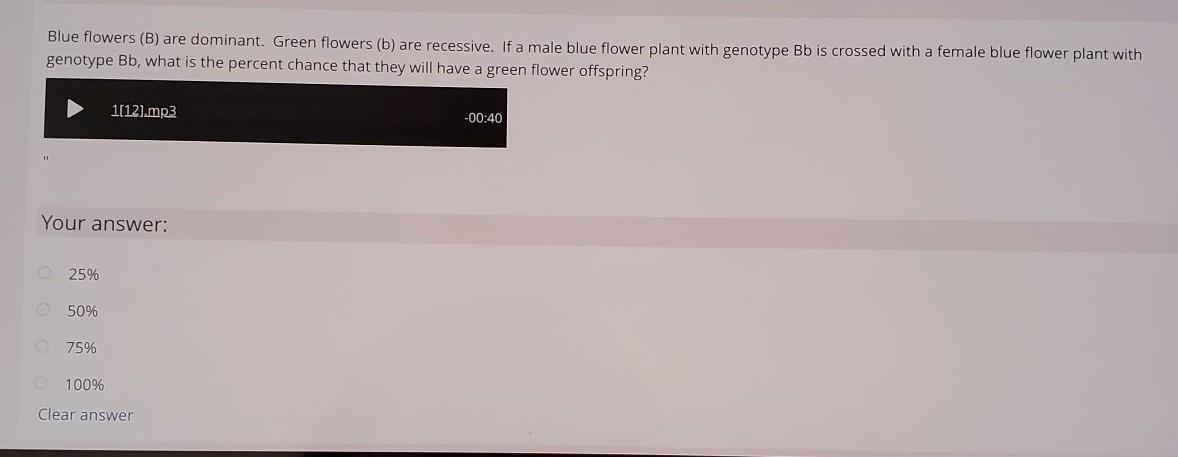
Answers
I hope this helps
Answer:
it is 25%
Explanation:
Answering so you can give the other person brainliest :D have a good day
Related Questions
HELP WHOEVER ANSWERS WILL BE MARKED BRAINLIEST!!!
Answers
Help help help !!!!!!
Answers
Answer:
3. 116.5 V
4. 119.6 V
Explanation:
3. Determination of the voltage.
Resistance (R) = 25 Ω
Current (I) = 4.66 A
Voltage (V) =?
V = IR
V = 4.66 × 25
V = 116.5 V
Thus, the voltage is 116.5 V
4. Determination of the voltage.
Current (I) = 9.80 A
Resistance (R) = 12.2 Ω
Voltage (V) =?
V = IR
V = 9.80 × 12.2
V = 119.6 V
Thus, the voltage is 119.6 V
How many particles are in 9.58 x 1023 of potassium sulfate (K2SO4)? (5.77 x 1047 particles)
Answers
Answer:
[tex]5.77x10^{47}particles.[/tex]
Explanation:
Hello!
In this case, since we are given 9.58x10²³ moles of potassium sulfate, it is possible to compute the particles according to the Avogadro's number:
6.022x10²³
As shown below:
[tex]9.58x10^{23}mol*\frac{6.022x10^{23}}{1mol}\\\\5.77x10^{47}particles.[/tex]
Best regards!
number of molecules in lithium oxide
4li2O
Answers
Answer:
Explanation:
Lithium is in group 1 so there's 1 outermost shell electron.
Oxygen is in group 6 so there's 6 outermost shell electron.
To react lithium with oxygen, we need 2 lithium and 1 oxygen
Each lithium transfer 1 electron to the oxygen and oxygen gain 2 electrons.
[tex]Li_{2}O\\[/tex] formed.
If there's 4 lithium oxide, 4 molecules will be there.
If you mean how many ATOMS are there, there will be 3×4=12 atoms.
Which element, when combined with fluorine, would most likely form an
ionic compound?
A. phosphorus
B. lithium
C. carbon
D. chlorine
Answers
I have an unknown initial volume of gas
held at a temperature of 115 K in a
container with a pressure of 6 atm. I
increase the temperature to 225 K and
decrease the pressure to 2,800 mmHg
and it causes the final volume of the gas
to be 29 L. What was the initial
volume?
Answers
Answer:
V₁ = 0.025 Liters = 25 ml
Explanation:
P₁ = 6 Atm P₂ = 7800mm/760mm/Atm = 0.01 Atm
V₁ = ? V₂ = 29 Liters
T₁ = 115 K T₂ = 225 K
P₁V₁/T₁ = P₂V₂/T₂ => V₁ = P₂V₂T₁/P₁T₂
V₁ = (0.01 Atm)(29L)(115K)/(6 Atm)(225K) = 0.025 Liters = 25ml
how many atoms are in 4.67 grams of sample of silicon
Answers
How many sulfur atoms are in 3.7 mol of SO2?
× 10 S atoms
Answers
Answer: 22.3 *10^23 S atoms
Explanation:
An aqueous solution contains
32.7% KCI (weight weight %)
How many grams of KCl are
contained in 100 g of this solution
Molar Mass
KCE: 74.55 g/mol
H20 18.016 g/mol
Answers
Answer:
32.7
Explanation:
I just did it and got it right
We obviously cannot wait for million years to determine the half-life of technetium, or even years to determine the half-life of plutonium. The half-life of a substance is determined using a Geiger counter, which is essentially a tube filled with an inert gas. When a particle decays and produces radiation, it briefly ionizes the gas and produces a detectable electric pulse, which is amplified and counted by a detector (before digital displays were available, you would hear clicks and see a needle register on a dial). Determine the formula for the half-life of a substance with an initial count of pulses and a count of pulses minutes later.
Answers
The question is incomplete, the complete question is;
We obviously cannot wait for 4 million years to determine the half-life of technetium, or even 87.7 years to determine the half-life of plutonium. The half-life of a substance is determined using a Geiger counter, which is essentially a tube filled with an inert gas. When a particle decays and produces radiation, it briefly ionizes the gas and produces a detectable electric pulse, which is amplified and counted by a detector (before digital displays were available, you would hear clicks and see a needle register on a dial). Determine the formula for the half-life of a substance with an initial count of No pulses and a count of N1 pulses M minutes later.
Answer:
M1/2 = 0.693/k
Explanation:
Given that the initial count rate is No pulses
The count rate after M minutes is N1
Then from;
A = Aoe^-kt
A= N1
Ao=No
I can now write;
N1 = Noe^-kM
The half life is the time taken for the activity of the radioactive nuclide to reach half its initial value. Hence;
N1/No =e^-kM = 1/2
Taking natural logarithm of both sides as shown below;
-kM1/2 = ln(1/2)
M1/2 = - (ln(1/2)/k)
M1/2 = 0.693/k
If 4 moles of sulfur reacts with 9.5 moles of oxygen, how many moles of oxygen would remain after the reaction ?
Answers
4* 23 g Na --------> 16 g O₂
9.5 g Na ------------> ?
Mass of O₂ = 9.5 * 16 / 4 * 23
Mass = 152 / 92
Mass = 1.6521 g of O₂
Molar mass O₂ = 16.0 g/mol
1 mole O₂ ------------ 16.0 g
? mole O₂ ------------ 1.6521 g
mole O₂ = 1.6521 * 1 / 16.0
≈ 0.10325 moles of O₂
hope that helped!
Calculate the percent composition (out of 100) by weight of potassium in potassium carbonate decahydrate.
Answers
Answer:
12.26%
Explanation:
MM K = 39.1 g/mol
MM K2CO3 . 10H2O = 2x39.1 + 12.01 + 3 x 15.99 + 10 x 18.02= 318.38 g/mol
% by weight = (MM K/MM K2CO3.10H2O ) x 100= (39.1/318.8) x 100=12.26 %
The percent composition by weight of potassium in potassium carbonate decahydrate is 13.98%
Let's represent the chemical formula of the compound as follows;
potassium carbonate decahydrate = CH₂0KO₁₃
The molecular mass of the compound can be calculated as follows:
Molecular mass of CH₂₀KO₁₃molecular mass = 12 + 20 + 39 + (16×13) = 279 g/molmass of potassium = 39 g
Therefore,
% mass of potassium = 39 / 279 × 100
% mass of potassium = 3900 / 279
% mass of potassium = 13.9784946237
% mass of potassium = 13.98 %
learn more on percentage composition here: https://brainly.com/question/3905173
Hyenas eat larger mammals like antelope and wildebeest, but will also eat lizards, birds, snakes, and insects. What type of teeth would you expect a hyena to have?
Answers
Answer:
Large Sharp Teeth
Explanation:
i got a 100 on my test!
Large Sharp Teeth !
i got it right on my quiz
Help plz now !
Which statement explains why a chemical equation must be balanced?
A. It must show the reactants and products on the correct sides of
the reaction arrow.
B. It must show that the mass of each element involved is conserved
by a chemical reaction.
C. It must show how each chemical formula is written to accurately
represent each substance.
D. It must show that coefficients and subscripts can be used in
chemical formulas.
Answers
Answer: B. It must show that the mass of each element involved is conserved by a chemical reaction.
Explanation:
According to the law of conservation of mass, mass can neither be created nor be destroyed. Thus the mass of products has to be equal to the mass of reactants. The number of atoms of each element has to be same on reactant and product side. Thus chemical equations are balanced.
The correct statement why a chemical equation must be balanced is It must show that the mass of each element involved is conserved by a chemical reaction.
Answer:
B. It must show that the mass of each element involved is conserved
by a chemical reaction.
Explanation:
a p e x, just took the quiz
Answer all the following questions by putting a circle around a letter A.B.C or D
1. Air contains about
A. 3% carbon (IV) oxide.
B. 4%noble gases.
C. 21%oxygen
D. 69% nitrogen
Answers
Answer:
C. 21%oxygen
Explanation:
Dry air contains 78.09% nitrogen, 20.95%oxygen, 0.93% argon, 0.04% carbon dioxide, and small amounts of other gases.
Which part of a calcium atom in the ground state is represented by the dots in its Lewis electron-dot diagram?
Answers
Answer:
There are two dots are present on the left side of calcium atom which represents the unpaired electrons present in the outermost shell. This structure is presented by Lewis in order to show the number of unpaired electrons in the atom.
Explanation:
Hardness is the measure of a mineral's resistance to scratching.
True
False
Answers
Answer:
The answer to the question is true.
2 CH3OH + 3 02 2 CO2 + 4H2O
What is the mass of oxygen (O2) that is required to produce 579 g of carbon dioxide (CO2)?
. Your answer should have three significant figures.
Answers
Answer:
632 g
Explanation:
2CH₃OH + 3O₂ → 2CO₂ + 4H₂OFirst we convert 579 g of CO₂ into moles, using its molar mass:
579 g CO₂ ÷ 44 g/mol = 13.16 mol CO₂Then we convert CO₂ moles into O₂ moles, using the stoichiometric coefficients:
13.16 mol CO₂ * [tex]\frac{3molO_2}{2molCO_2}[/tex] = 19.74 mol O₂Finally we convert O₂ moles into grams, using its molar mass:
19.74 mol O₂ * 32 g/mol = 632 gwhat is a 7 letter word that starts with f: Ancient plants and animals formed into rocks.
Answers
Answer:
Fossils?
Explanation:
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. An industrial chemist studying this reaction fills a flask with of sulfur dioxide gas and of oxygen gas, and when the mixture has come to equilibrium measures the partial pressure of sulfur trioxide gas to be . Calculate the pressure equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. Round your answer to significant digits.
Answers
Answer:
The answer is "[tex]\bold{4.97 \times 10^{-2}}[/tex]"
Explanation:
Please find the complete question in the attached file.
Equation:
[tex]2SO_2+O_2 \leftrightharpoons 2SO_3[/tex]
at [tex]t=0 3.3 \ \ \ \ \ \ \ \ \ \ 0.79[/tex]
at equilibrium [tex]3.3-p \ \ \ \ \ \ \ \ \ \ 0.79 - \frac{P}{2} \ \ \ \ \ \ \ \ \ \ \ \ P[/tex]
[tex]p= 0.47 \ \ atm\\\\SO_2=3.3-0.47 = 2.83 \ \ atm\\\\O_2= 0.74 -\frac{0.47}{2}=0.74-0.235=0.555 \ atm\\\\K_P=\frac{[PSO_3]^2}{[PSO_2]^2[PO_2]}\\\\[/tex]
[tex]=\frac{0.47^2}{2.83^2\times 0.555}\\\\=4.97 \times 10^{-2}[/tex]
If you have 9.56 moles of aluminum oxide how many moles of sodium oxide could you produce?
Answers
Answer:
The answer is 28.68 moles
Explanation:
Hope this helped Mark BRAINLEST!!!
What is the molar mass of H2CO3?
(Molar mass of H = 1.0079 g/mol; C = 12.010 g/mol; O = 15.999 g/mol)
Answers
Answer:
molar mass of H2CO3 = 72.0228
Answer:
62.02 g/mol
Explanation:
Took the test and got it right. if you have enough faith in brainly youll choose the right answer
help someone i need this
Answers
which property is a property of water
Answers
Answer: Polarity, Boiling and Freezing Points, Specific Heat Capacity, Density, Surface Tension, Heat of Vaporization and Vapor Pressure, Capillary Action, Solid State (Ice), Liquid State (Liquid Water), Gas State (Steam),
Explanation: Water molecules are polar, so they form hydrogen bonds. This gives water unique properties, such as a relatively high boiling point, high specific heat, cohesion, adhesion, and density.
How many grams of AlCl3 will be produced if 3.85 moles of Al react? Al + ___Cl2 → ___AlCl3
Answers
Answer:
513.975g
Explanation:
I'm assuming that the question has Aluminium and Chlorine as the question is not clear.
1. Balance the equation
2 Al + 3 Cl2 → 2AlCl3
2. Use the mole = mass/mr equation and molar ratio to find the mass of AlCl3
133.5 x 3.85 = 513.975g
Please help ASAP! Please and thank you have a great and blessed day!,
Answers
What is produced when concentrated aqueous sodium chloride is electrolysed?
Answers
You’re welcome!! Have a good day/night :)
which statement explains this observation?
Answers
Answer:
should be D. Hope this helpsss!
Which statement below best describes a volatile liquid?
Question 17 options:
1.A liquid that doesn't evaporate under atmospheric pressure
2.A liquid that evaporates slowly at low temperatures
3.A liquid that evaporates rapidly at low temperatures
4.A liquid that requires a large amount of energy to evaporate
Answers
Answer:
A liquid that evaporates rapidly at low temperatures
Explanation:
Answer:
A liquid that evaporates rapidly at low temperatures.
Explanation:
A volatile liquid such as gasoline evaporates easily. Its chemicals aren't held together as strongly, and room temperature is often enough to break intermolecular bonds and evaporate them.
PF
help, if anyone knows the correct answer??