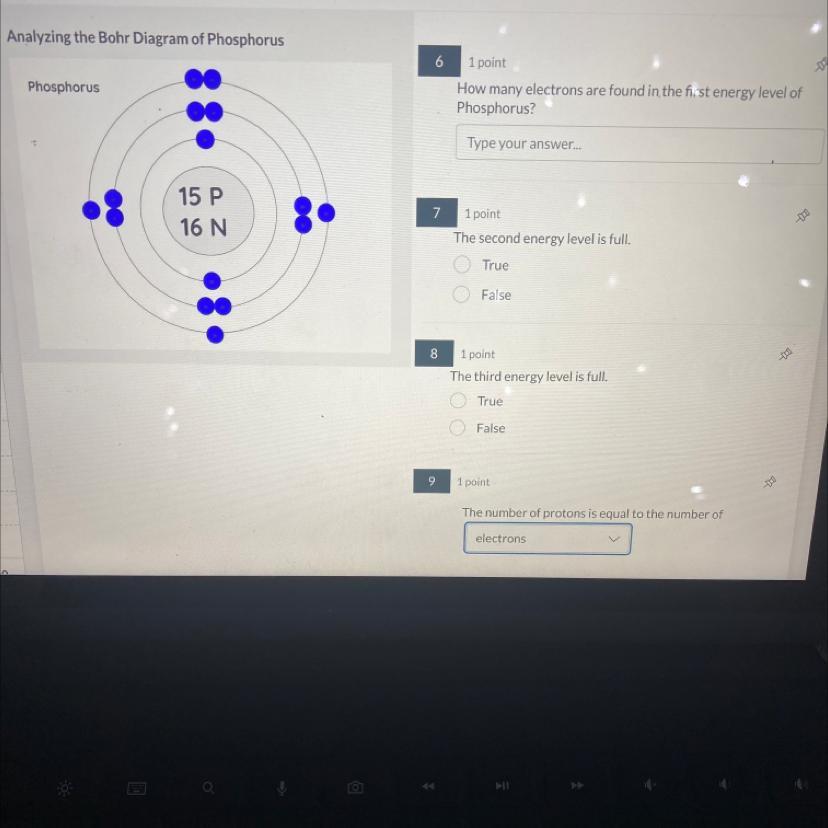
Answers
Answer:
15 electrons
Explanation:
Related Questions
2. Infer what is meant by an amorphous solid.
Answers
Answer:
Amorphous solid is a form of solid
Explanation:
Amorphous solid is a solid in which atoms are not arranged in a definite geomatric pattern for eg. glass,paper etc
0.10 mm kmno4 has an absorbance maximum of 0.26 at 525 nm in a 1.000-cm cell. find the molar absorptivity and the concentration of a solution whose absorbance is 0.52 at 525 nm in the same cell. watch your units.
Answers
Molar absorptivity and the concentration of the solution whose absorbance is 0.52 at 525nm is 2600M-1 cm-1 and 0.20mM.
Molar absorbance is the measure how strongly a chemical species absorbs at given wavelength of light. Beers law relates the absorbance of a solution to its molar concentration. Beers law states that the light that absorbs by the solution is directly proportional to the concentration of the substance absorbing the light.
Absorbance and concentration are related to each other using the path length of light and molar absorptivity of the substance. Molar absorptivity of a substance is given by,
A= E.I.C
0.26= 1.00cm .e . 0.10 . 10-3 M
e= 0.26 / 0.10 *10-3 M.cm
= 2600M-1cm-1
In the same way we have to calculate the concentration of the another sample using path length and molar absorptivity which are same for both the samples .
Calculating the relation, we got the concentration of 0.2mM.
To know more about Beers law please visit:
https://brainly.com/question/18205869
#SPJ4
1. in a few sentences define each of the three main classes of isomers below. configurational isomers can be further classified as either enantiomers or diastereomers and should also be discussed. your descriptions should enable your peers to quickly and easily compare and contrast the differences between the different classes of isomers. a. constitutional isomers (or structural isomers) b. conformational isomers c. configurational isomers(or stereoisomers) i. enantiomers ii. diastereomers
Answers
Configurational isomers are stereoisomers that can't be transformed into each other via rotating the molecule around a single bond. those configurational isomers may be found in kinds such as geometrical isomers and optical isomers.
Enantiomers are pairs of compounds with precisely equal connectivity however contrary to three-dimensional shapes. Enantiomers aren't the same as every different; one enantiomer can not be superimposed on the alternative. Enantiomers reflect images of every different.
Diastereomers are a kind of stereoisomer. Diastereomers are described as non-mirror pictures, non-same stereoisomers. hence, they arise whilst two or greater stereoisomers of a compound have specific configurations at one or greater of the equal stereocenters and aren't replicate pics of every different
Learn more about configurational isomers here:-https://brainly.com/question/26298707
#SPJ4
Gravity is a (n) _____________ between objects and depends on an object's size and their distance apart.
Answers
Gravity is a (n) force between objects and depends on an object's size and their distance apart.
Gravitational force is the attractive that exist between all object with mass an object with mass attracts another object with mass the magnitude of the force is directly proportional to the masses of the two objects and inversely proportional to the square of the distance between the two objects
Know more about objects
https://brainly.com/question/27786016
#SPJ1
a student needs to prepare 250 ml of a 0.800 m aqueous solution of sucrose, c12hz2011 (aq), which is used frequently in biological experiments 200 ml 250 ml 150 200 100 150 100 250 ml which type of glassware should be used to make this solution (assuming that the accuracy of the concentration is important)? 0 a how should the correct amount of solute be obtained? measure out x cm of sucrose with a ruler: measure out x g of solid sucrose on a balance. measure out x mol of solid sucrose on a molemeter based on the selected correct unit; what is the value of x?
Answers
The type of glassware that students need to use to prepare 250 ml of a 0.800 m aqueous solution of sucrose is a volumetric flask and the correct amount of solute to use is 68.46g.
It is always recommended to use volumetric flask when you make sucrose solution, as accuracy is very important. Other glassware can be used to make water measurements more accurate, but it is always better to use a volumetric flask to this stock solution. and the correct amount of solute to use can be calculate as follow:
In this case, sucrose is a solid compound, so the best way to measure it is to use a balance and weigh the required mass based on the concentration of the solution. Therefore, it is best to measure grams of solute on a scale.
The amounts required are:
M = n/V
n = M*V
n = 0.800 * 0.250 = 0.2 moles
To get the mass:
m = n*MM
The molar mass of sucrose is 342.3 g/mol.
m = 0.2 * 342.3
m = 68.46g
Finally, after we have weighed the mass of sucrose we need, so all that remains is to add the solute to the volumetric flask and add water. In this way, sucrose mixes with water into a solution. Then, to make the solution 0.800M, the volume should be 250mL.
learn more about sucrose here https://brainly.com/question/15074456
Write a balanced equation for the complete oxidation reaction that occurs when methanol (ch3oh) burns in air.
Answers
The balanced equation for the complete oxidation reaction that occurs when methanol CH₃OH burns in air is 2CH₃OH + 3O₂ → 2H₂O + 4CO₂.
When Methane CH₃OH reacts with O₂, it get oxidized and CO₂ and H₂O are formed.
The chemical equation of the oxidation of methane when it burns in the air is,
CH₃OH + O₂ → H₂O + O₂
In order to balance the reaction, we should start first with carbon, then Hydrogen and at the end, we will balance oxygen atoms on both the sides of the reaction,
So, the balanced chemical equation becomes,
2CH₃OH + 3O₂ → 2H₂O + 4CO₂
So, in this reaction, Carbon is getting oxides as it is becoming carbon dioxide by the addition of oxygen into it and Oxygen itself is getting reduced.
To know more about oxidation, visit,
https://brainly.com/question/25886015
#SPJ4
octyl formate is an ester that has the flavor of oranges. it can be synthesized from an alcohol and a carboxylic acid. what alcohol is needed to make octyl formate? choose... what carboxylic acid is needed to make octyl formate?
Answers
Octanol alcohol is needed to make octyl formate and Formic Acid carboxylic acid is needed to make octyl formate.
The organic substance 1-octanol, commonly referred to as octan-1-ol, has the chemical formula CH3(CH2)7OH. The alcohol is fatty. In general, octanols refer to a wide variety of different isomers. In the industrial setting, octanol is mostly generated by oligomerizing ethylene with triethyl aluminum, followed by oxidation of the resulting alkylaluminium products. The Ziegler alcohol synthesis is the name of this method.
An organic acid with a carboxyl group connected to an R-group is referred to as a carboxylic acid in organic chemistry. R stands for the alkyl, alkenyl, aryl, or other group, and the typical formula of a carboxylic acid is RCOOH or RCO2H. It is common to find carboxylic acids.
learn more about octanol here
https://brainly.com/question/28168707
#SPJ4
please help when ever it lets me give brainliest i will give it
Answers
Answer:
it's sample c yh it is true
explain how the direction of electron flow in a voltaic cell is consistent with what you would predict from the activity series
Answers
In voltaic cells, electrons flow from the negative electrode (anode) to the positive electrode (cathode).
Electrons are moved from one species to another in redox reactions. When a reaction occurs spontaneously, energy is released that can be put to good use. The process must be divided into the oxidation reaction and the reduction reaction in order to capture this energy. A wire is used to move the electrons from one side of the reactions to the other after they have been placed into two separate containers. A voltaic/galvanic cell is produced as a result.
To know more about voltaic/galvanic cell, visit;
brainly.com/question/28051381
#SPJ4
1. what is an aquifer? a. a water purification system b. water distribution system c. geological formation that stores water d. a drinking water solution
Answers
An aquifer is a geological formation that stores water. they are higher than the surrounding terrain, which can lead to springs or artesian wells that are in motion. An aquifer can be a well, spring, lake ,etc.
What is an aquifer?Water collects in geological formations called aquifers, frequently at great depths in the earth. Some aquifers are higher than the surrounding terrain, which can lead to springs or artesian wells that are in motion.
Hence, An aquifer is a geological formation that stores water. they are higher than the surrounding terrain, which can lead to springs or artesian wells that are in motion.
to know more about aquifer visit
https://brainly.com/question/14916917
#SPJ4
pls answer question will mark brainliset tyty question is in the picvture
Answers
Answer:
d
Explanation:
exotic species, took this last week.
Mass has to do with how many particles
of matter are in something. Something
with more particles of matter will have...
Answers
The law of definite proportions agrees with Dalton atomic theory.
What is Dalton atomic theory?
It state that all matters is made of very tiny particles called atom. atoms are individual particles which can not be created or be destroyed in a chemical reactions. Atoms of given elements are identical in mass and chemical properties. Atoms of
different elements have different masses and chemical properties.
The law of definite proportions also known as proust's law ,state that a chemical compound contain the same proportion of elements by mass.this law is one of the stoichiometry .
Thus ,
This is the reason why it is agrees with dalton atomic theory.
To know more about Dalton atomic theory click-
https://brainly.com/question/13157325
#SPJ1
27.A full bag of trash is placed in a trash compactor. After compacting, which of the following properties of the bag of trash have changed?1. Mass2. Volume3. DensitySelect one:a. 1 onlyb. 1 and 2 onlyc. 2 and 3 onlyd. 2 only
Answers
ANSWER
option D
EXPLANATION
When a full bag of trash is placed in a compactor, the compactor will compress the bag so as to allow more trash to fill in. This is done by increasing the capacity of the trash.
Therefore, the volume of the trash bag will changed
Hence, the correct answer is option D
A 35. 0 ml portion of 0. 255 m nitric acid is added to 45. 0 ml of 0. 328 m mg(no3)2. What is the concentration of nitrate ion in the final solution?.
Answers
The concentration of nitrate ion in the final solution is 0.48 M.
An abundance of a constituent divided by the total volume of a mixture is known as concentration. There are various types of concentrations: Mass, molar, number, and volume. The quantity of a solute found in the given amount of solution is the concentration of the substance. Concentration is expressed in terms of molarity.
Molarity means the number of moles per litre. The unit symbol is mol / L or mol / dm³ in the SI unit.
To calculate the concentration of the solution,
[tex]C_{1}V_{1} + n C_{2}V_{2} = C * total volume[/tex] → 1
where, [tex]C_{1}[/tex] = concentration of the starting solution
[tex]C_{2}[/tex] = concentration of the desired solution
[tex]V_{1}[/tex] = volume of the starting solution
[tex]V_{2}[/tex] = volume of the desired solution
As per the question,
[tex]C_{1}[/tex] = 0.255 M[tex]C_{2}[/tex] = 0.328 M [tex]V_{1}[/tex] = 35.0 ml = 0.035 L [tex]V_{2}[/tex] = 45.0 ml = 0.045 LSubstitute the values in 1,
⇒ [tex]C_{1}V_{1} + n C_{2}V_{2} = C * total volume[/tex]
⇒ 0.255 × 0.035 + 2 × 0.328 × 0.045 = C × 0.08
⇒ 0.008925 + 2 × 0.01476 = C × 0.08
⇒ 0.008925 + 0.02952 = C × 0.08
⇒ C × 0.08 = 0.038445
⇒ C = 0.038445 / 0.08
⇒ C = 0.4805625
Therefore, 0.48 M is the concentration of the Nitrate ion in the final solution.
To know more about the concentration of the solution refer to:
https://brainly.com/question/26255204
#SPJ4
the number 207.2 in the box labeled Pb on the periodic table represents..?
1) the number of electrons and neutrons
2)the number of electrons in a neutral atom
3) the average atomic mass
4)the number of protons, neutrons, and electrons
5)the atomic matter
Answers
The properties of the given element, Pb, is as follows:
the number of electrons is 82 and the neutrons is 125the number of electrons in a neutral atom is 82the average atomic mass is 207.2the number of protons = 82, neutrons = 125, and electrons = 82the atomic matter is solid.What is the periodic table?The periodic table is an arrangement of the elements in increasing order of atomic number.
The atoms of elements are composed of three sub-atomic particles;
electrons- negatively charged particlesprotons - positively charged particlesneutrons - neutral particlesThe element Lead, Pb, is a metallic element found in the periodic table and has an atomic number of 82 and an average atomic mass of 207.2
Learn more about protons and electrons at: https://brainly.com/question/6258301
#SPJ1
The dog has a mass of 57kg and the boy has a mass of 48 kg. Who has more kinetic energy?
Answers
Answer:
The Dog
Explanation:
The more mass something has the more kinetic energy it has in it.
Question 4
Calculate the average atomic mass of the fictional element "X" by using the
information in the table below:
Isotope
X-16
X-17
X-18
Abundance (%)
99.76
0.04
0.20
Atomic Mass
15.9949
16.9991
17.9992
Answers
The average atomic number will be 1616.29.
The Average Atomic Mass = (Mass of Isotope 1 x Fractional Abundance of Isotope 1) + (Mass of Isotope 2 x Fractional Abundance of Isotope 2)+ (Mass of Isotope 3x Fractional Abundance of Isotope 3)
Given,
X-16
X-17
X-18
Abundance (%)
99.76
0.04
0.20
Atomic Mass
15.9949
16.9991
17.99
Average Atomic mass= (15.9949x99.76)+(16.9991x0.04)+(17.9992x0.20)
=1616.29
Therefore, average atomic mass of the fictional element "X" is 1616.29
To know more about Atomic mass do visit
https://brainly.com/question/13671163
#SPJ1
16.Sulfur has 16 electrons. After it bonds with Magnesium it acquires a 2- charge. How many electrons does the S2- ion contain?Select one:a. 2b. 8c. 16d. 18
Answers
18 electrons.
Explanations:According to the question, we have that sulphur has 16 electrons and after bonding with magnesium acquired a 2-charge. The sulphur ion S2- shows that sulphur has gained 2electrons to become stable.
Amount of electron S2- have = 16 electron + 2electrons
Amount of electron S2- have = 18 electrons
Hence the number of electrons S2- ion have is 18 electrons.
Molecules of gas in the air absorb
light of shorter wavelengths in the
range of 450-495nm is an example
of?
Answers
Molecules of gas in the air absorb light of shorter wavelengths in the range of 450-495nm is an example of quantitative data.
Quantitative data is defined as the data that can be counts in number or can be measured in numbers. There are two main types of quantitative data : discreate data and the continuous data. examples are age , date, distance , weight etc. so, the countable and measurable quantities in numbers is quantitative data. Wavelength here is measured in numbers . so, wavelength is the quantitative data.
Thus, Molecules of gas in the air absorb light of shorter wavelengths in the range of 450-495nm is an example of quantitative data.
To learn more about Quantitative data here
https://brainly.com/question/14810759
#SPJ1
Copper was one of the earliest metals used by humans, because it can be prepared from a wide variety of copper minerals, such as cuprite (cu2o), chalcocite (cu2s), and malachite [cu2co3(oh)2]. Balance the following reactions for converting these minerals into copper metal. Place a coefficient in each gray box.
Answers
Will be the balanced chemical reaction.
A chemical reaction is a procedure that causes one group of chemical components to change chemically into another. Chemical reactions, which can frequently be described by a chemical equation, traditionally include changes that only involve the positions of electrons in the formation and dissolution of chemical bonds between atoms, with no change to the nuclei (no change to the elements present). The study of chemical reactions involving unstable and radioactive elements, where both electronic and nuclear changes may take place, is known as nuclear chemistry.
To know more about chemical reaction, click here,
brainly.com/question/11231920
#SPJ4
Where do convection currents happen?(1 point)
Responses
A.crust
B.core
C.mantle
D.inner core
Which statement is correct about the movement of plates that sit on top of the mantle? (1 point)
Responses
A.They regularly move because of the convection currents of magma.
B.They never move because they are connected to Earth’s crust.
C.They never move because they are connected to mountains.
D.They regularly move because of the convection currents of oceans.
Which statement is correct about the flow of thermal energy inside Earth?(1 point)
Responses
A.Energy flow inside the mantle causes magma to sink deeper.
B.Energy flow inside the crust causes magma to rise.
C.Energy flow inside the crust causes magma to sink deeper.
D.Energy flow inside the mantle causes magma to rise.
Which is a result of boundary plates colliding or sliding past each other? (1 point)
Responses
A.valleys form
B.earthquakes occur
C.mountains form
D.volcanoes erupt
Answers
The convection currents happen in the mantle, option C.
The statement is correct about the movement of plates that sit on top of the mantle is option D.They regularly move because of the convection currents of oceans.
The statement that is correct about the flow of thermal energy inside Earth is optionB. Energy flow inside the crust causes magma to rise.
The result of boundary plates colliding or sliding past each other is option B. earthquakes occur
How can be the Convection currents described?Convection currents can be identified in the Earth's mantle wherby the materials that are been heated which is the mantle material will be rising from deep inside the mantle, then the cooler mantle material will definitely sinks which will bring about the convection current.
It should be noted that the Transform boundaries which is as a result of the sliding of the plates could results in shallow earthquakes.
Learn more about convection currents here: https://brainly.com/question/20295382
#SPJ1
Answer: Where do convection currents happen?
Responses= mantle
Which statement is correct about the movement of plates that sit on top of the mantle= They regularly move because of the convection currents of magma.
Which statement is correct about the flow of thermal energy inside Earth= Energy flow inside the mantle causes magma to rise.
Which is a result of boundary plates colliding or sliding past each other= earthquakes occur
Explanation: I took the test
Ammonia produced from the Haber process is responsible for feeding over a quarter of
the world’s population. Using this reaction equation, answer the question below.
N₂ + 3H₂ ⇒ 2NH₃
1. If you had 3 moles of N2 and an excess of H2, how many moles of NH3 would be produced?
2. How many moles of N2 are needed to react completely with 12 moles of H2?
3. If you had 30 molecules of H2 and an excess of N2, how many molecules of NH3 would be produced?
Answers
1. 2 moles of nitrogen reacting with an excess of hydrogen will produce moles of ammonia6 moles [tex]NH_3[/tex].
2. The number of moles of nitrogen needed to completely react with 12 moles of hydrogen is 4 moles.
3. 30 molecules of hydrogen reacting with an excess of nitrogen will produce 20 molecules of ammonia.
Stoichiometric problemThe balanced equation of the reaction between hydrogen and nitrogen to produce ammonia is as follows:
[tex]N_2 + 3H_2 -- > 2NH_3[/tex]
This means that the mole ratio of [tex]N_2[/tex] to [tex]H_2[/tex] to [tex]NH_3[/tex] is 1:3:2.
If there are 3 moles of [tex]N_2[/tex] an excess of [tex]H_2[/tex]. The mole ratio of [tex]N_2[/tex] to [tex]NH_3[/tex] is 1:2. This means that for every 1 mole [tex]N_2[/tex] that reacts, 2 moles of [tex]NH_3[/tex] are produced.1 mol [tex]N_2[/tex] = 2 mol [tex]NH_3[/tex]
3 mol [tex]N_2[/tex] = 3 x 2
= 6 moles [tex]NH_3[/tex]
The mole ratio of [tex]N_2[/tex] and [tex]H_2[/tex] is 1:3.3 mol [tex]H_2[/tex] = 1 mol [tex]N_2[/tex]
12 mol [tex]H_2[/tex] = 12 x 1/3
= 4 moles [tex]N_2[/tex]
The mole ratio of [tex]H_2[/tex] and [tex]NH_3[/tex] is 3:2.3 mol [tex]H_2[/tex] = 2 mol [tex]NH_3[/tex]
30 mol [tex]H_2[/tex] = 30 x 2/3
= 20 moles [tex]NH_3[/tex]
In other words, the problem is solved from the balanced equation of the reaction.
More from stoichiometric problems can be found here: https://brainly.com/question/28297916
#SPJ1
True or False: A neutrally charged atom has an equal number of protons and electrons.
Answers
Answer:
True
Explanation:
Answer:
True
Explanation:
A neutral atom must have an equal amount of protons as electrons.
What are electrolytes and why are they important? (1 pt)
Unit 4 lesson 4-6
Answers
Minerals with an electric charge called electrolytes are found in your body. They can be found in various bodily fluids such as urine, tissues, and blood.
Why are electrolytes important?
Our body contains electrolytes, which are minerals with an electric charge. Our blood, urine, tissues, and other bodily fluids all contain them.Electrolytes are crucial because they aid in:
Ensuring that our body is adequately hydrated.Maintain a pH balance in our body.Transfer nutrition to our cells.Clear our cells of wastes.Make sure our heart, brain, muscles, and nerves all function properly.The many electrolytes include sodium, calcium, potassium, chloride, phosphate, and magnesium. They come from the foods we consume and the liquids we drink.
The body's electrolyte levels might get too high or too low when the body's water balance changes. The amount of water we consume and lose should be equal. If something throws off this equilibrium, we can be dehydrated or have too much water on our body (overhydration).
The water balance can be thrown off by a number of factors, including some medications, nausea, diarrhea, perspiration, and liver or kidney issues.
We can regulate the imbalance with the aid of treatment. Additionally, the cause of the imbalance must be determined and addressed.
To learn more about electrolytes from the given link
https://brainly.com/question/17089766
#SPJ1
A 35.40 gram hydrate of sodium carbonate, Na2CO3•nH2O, is heated to a constant mass. Its final weight is 30.2 g. What is formula for the hydrate?A. Na2CO3∙1H2OB. Na2CO3∙2H2OC. Na2CO3D. Na2CO3∙3H2O
Answers
First, we have to calculate the molecular weights of each molecule:
[tex]\begin{gathered} Na_2CO_3\text{ : 23*2+12+16*3= 106 g/mol} \\ H_2O\text{ : 1*2+16= 18 g/mol} \end{gathered}[/tex]Then, we have to calculate the number of grams of water. We can calculate them because the process of evaporation lets us know the water amount that was retired:
[tex]g\text{ H}_2O\text{ = 35.40 g - 30.2 g=5.2 g H}_2O[/tex]Then, we're gonna convert the grams of sodium carbonate alone (30.2 g) and the grams of water to moles:
[tex]\begin{gathered} 30.2\text{ g Na}_2CO_3\text{ * }\frac{1\text{ mol}}{106\text{ g}}=\text{ 0.2849 mol Na}_2CO_3\text{ }\approx0.3\text{ mol Na}_2CO_3 \\ \\ 5.2\text{ g H}_2O\text{ * }\frac{1\text{ mol}}{18\text{ g}}=\text{ 0.288 mol H}_2O\text{ }\approx\text{ 0.3 mol H}_2O \end{gathered}[/tex]It means that the mole relation is 1:1 approx, as it is the same amount for both. Then, the formula is going to be:
[tex]Na_2CO_3\text{ . 1H}_2O[/tex]It means that the answer is A.
A gas has a temperature of 34.9 degrees C and a volume of 70.0 L. If the temperature increases to 86.8 degreesC , what is its final volume ?
Answers
Answer
Its final volume is 81.8 L
Explanation
Given;
Initial temperature, T₁ = 34.9°C = (34.9 + 273.15 K) = 308.05 K
Initial volume, V₁ = 70.0 L
Final temperature, T₂ = 86.8°C = ( 86.8 + 273.15 K) = 359.95 K
What to find:
The final volume at 86.8°C.
Step-by-step solution:
The final volume, V₂ can be calculated using Charle's law formula below:
[tex]\begin{gathered} \frac{V_1}{T_1}=\frac{V_2}{T_2} \\ \\ \Rightarrow V_2=\frac{V_1T_2}{T_1} \end{gathered}[/tex]Putting the values of the given parameters into the formula, we have
[tex]V_2=\frac{70.0L\times359.95K}{308.05K}=81.8\text{ }L[/tex]Hence, if the temperature of the gas increases to 86.8 degrees C, its final volume is 81.8 L
Answer:
81.8 L
Explanation:
Ideal gas law:
PV = nR T
V = nR/P * T since nr and P are constant , this becomes
V = k T where k is a proportionality constant ====> then: V/T = k
so:
V1/T1 = V2/T2 ( NOTE : T must be in K)
V2 = T2 ( V1/T1) = ( 86.8 + 273.15) ( 70.0) /(34.9 + 273.15) = 81.8 L
. what, if anything, would be different about the product of the reaction if you started with z-stilbene instead of e-stilbene?
Answers
Answer: Yes the product will be different. If we use z - stilebene we would obtain trans dibromide.
Explanation:
Just as you had started with a molecule with two phenyl substituents on the same side, the product has the configuration on each stereocenter the same as the other (R,R; S,S).
If you rotate the central
C-C bond by about 120∘ you can compare these more closely.
Had you started with (E)-stilbene instead of (Z)-stilbene, you would have gotten meso (1S, 2R) and meso (1R, 2S) isomers instead.
What measuring tool would you use to measure the area of a
football field? Taking into account the tool you just chose, to what
decimal place would you record your answer? (make up some
numbers that represent the dimensions of the field and use them to
explain your answer)
Answers
Measuring tool would you use to measure the area of a football field is used a tender wheel
If you need to measure much longer lengths for example the length of a football pitch then you could use a trundle wheel then you use it by pushing the wheel along the ground and it clicks every time it measures one meter and to measure the football field area then
Area = length×breadth
Area = 90m×45m
Area = 4.05m²
Know more about area
https://brainly.com/question/19896101
#SPJ1
As the pressure of a system increases, what happens to the volume?
Answers
To answer this, we use Boyle's Law:
States that, at constant temperature, the pressure P of a gas varies inversely with its volume V.
So, if the pressure increases, the volume will decrease.
Answer: The volume decreases.
the following initial rate data are for the reaction of hypochlorite ion with iodide ion in 1 m aqueous hydroxide solution: experiment , m , m initial rate, 1 0.00381 0.00794 2 0.00381 0.0159 3 0.00762 0.00794 4 0.00762 0.0159 complete the rate law for this reaction in the box below. use the form , where '1' is understood for or and concentrations taken to the zero power do not appear.Don't enter 1 for m or n Rate = From these data, the rate constant is M-1s-1. Submit Answer.
Answers
The rate law in general form for this reaction is:
rate = k[A]^m x [B]^n
and we want to determine m and n.
Notice the experiments conducted involved verifying the concentration of one of the reactants while keeping the other constant. By comparing two experiments in this way and noticing the effect on the reaction rate, one can deduce the order with respect to that reactant.
Take experiments 1 and 2 :
Doubling the concentration of OH⁻ while keeping (CH₃)₃CBr constant had no effect on the initial rate.
This implies the order with respect to [OH⁻] is zero.
From experiments 1 and 3 we see that doubling the [ (CH₃)₃CBr ] while keeping [OH-] the same doubles the initial rate.
We now have the orders of the two reactants, and the rate law will be:
rate = k [ (CH₃)₃CBr ] [ OH⁻]⁰ = k [ (CH₃)₃CBr ]
We could use another comparison to check our answer. For example comparing 3 and 4, maintaining [ (CH₃)₃CBr ] constant and doubling [ OH⁻] has no effect on the rate which confirms the order respect to [ OH⁻] is zero.
The rate constant we can determine it from any of the experiment by solving for k, for example from experiment 1:
7.14 x 10⁻³ M/s = k x 0.626 M ⇒ k = 0.011 s⁻¹
Therefore, the rate constant of the following reaction is 0.011 s⁻¹.
To know more about rate constant, refer: https://brainly.com/question/20305922
#SPJ4