How do you determine the bond between two elements?
Answers
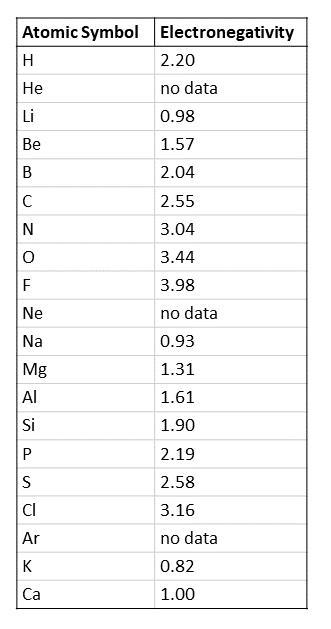
Step 1: Look at the atoms in the molecule and look up their electronegativity. Use the following table attached below to look up the electronegativities of the first 20 elements.
Step 2: Calculate the difference between the electronegativities of the atoms across each bond by subtracting the lower electronegativity from the higher electronegativity.
Step 3: Determine the bond type based on the differences in electronegativities. Ionic bonds have differences in electronegativity of 1.7 and higher. Polar-covalent bonds have differences in electronegativity of between 1.7 and 0.4. Covalent bonds have differences in electronegativity of 0.4 or lower.
To know more about electronegativity visit here ; https://brainly.com/question/17762711?referrer=searchResults
#SPJ4
Related Questions
The cell shown below could be a(n)
Answers
Answer:
the answer is bacteria cell
hexane burns according to the following equation: 2c6h14(g) 19o2(g) 12co2(g) 14h2o(g) what volume of co2 forms when 8.00 l of hexane burn, assuming the two volumes are measured under the same conditions? what volume of oxygen will be needed?
Answers
8.00L of hexane burns to produce 48.00L of CO2, if the two volumes are measured under identical circumstances. It takes 76.00L of oxygen.
Considering the response:Calculation:
C₆H₁₄ + 19/2O₂ → 6CO₂ + 7H₂O
where 6 moles of CO2 and 7 moles of H2O are produced when 1 mole of hexane combines with 19/2 moles of oxygen.
Volume is directly proportional to moles of gas according to Avogadro's law.6 moles of CO2 are created when 1 mole of hexane reacts with 8.00 liters, and their volume is as follows:
8.00L ₓ 6 = 48.0L
48.00L of CO₂ are formed.Again, 19/2 moles of oxygen are required for a full combustion of hexane. You need: Given that you have 8.0L of hexane.
8.00L × (19/2) =76.00L
76.00L of O₂ are neededTo know more about hexane burns visit:-
https://brainly.com/question/12216365
#SPJ4
a flask has a mass of 78.23 g when empty and 593.63 g when filled with water. when the same flask is filled with concentrated sulfuric acid, h2so4, its mass is 1026.57 g. what is the density of concentrated sulfuric acid in g/cm3? (assume water has a density of 1.00 g/cm3 at the temperature of the measurement.)
Answers
the density of concentrated sulfuric acid is 1.84 g/mL when a flask has a mass of 78.23 g when empty and 593.63 g when filled with water and later with sulfuric acid whose mass is 1026.57 g.
Given mass of flask mf = 78.23 g
Mass of flask when filled with water mfw = 593.63 g
Mass of water mw = mfw - mf = 593.63 - 78.23 = 515.4g
Volume of water Vw = Volume of acid Vc = 515.4mL
Mass of flask when filled with acid mfa = 1026.57 g
Mass of acid ma = mfa-mf = 1026.57 - 78.23 = 948.34g
Density = mass(ma)/volume of acid (Va)
= 948.34/515.4 = 1.84 g/mL
To learn more about sulfuric acid click here https://brainly.com/question/28513840
#SPJ4
The structural formula of an ester is CH 3 CH 2 COOC 2 H 5 . Write the name and structural formula of
products obtained when it is hydrolysed with dil. HCl.
Answers
Answer:
The structural formula of the ester is CH3CH2COOC2H5. When this ester is hydrolyzed with dilute hydrochloric acid (HCl), it will produce two products: ethanol (CH3CH2OH) and acetic acid (CH3COOH). The structural formulas of these products are shown below:
Ethanol: CH3CH2OH
Acetic acid: CH3COOH
Note that in a hydrolysis reaction, an ester is typically broken down into a carboxylic acid and an alcohol by the addition of water. In this case, the hydrolysis of the ester with dilute HCl results in the production of ethanol and acetic acid.
entropy never decreases in a spontaneous process. give an example to support this statement.
Answers
The perfect example for entropy of a spontaneous reaction would be the transfer of energy by heat from colder bodies to hotter bodies.
Spontaneous reactions are those chemical or biological reactions that usually take place without the influence of external factors. Non- Spontaneous reactions are those chemical reactions that generally requires an energy input to proceed or that cannot take place without the influence of external factors.
The transfer of energy by heat from colder bodies to hotter bodies is a spontaneous process in which the entropy of the system of bodies increases. The melting of an ice cube placed in a room causes an increase in the entropy of the room.
A spontaneous reaction may also involve an increase or decrease in enthalpy, it may involve an increase or decrease in entropy, but it will always involve a decrease in free energy that is a negative ΔG.
To know more about entropy and Spontaneous reaction
https://brainly.com/question/13146879
#SPJ4
What is the noble gas electron configuration for iodine
Answers
Answer:
See explanation and Image
Explanation:
Chemical element iodine has the atomic number 53 and the letter I in its symbol. of the stable halogens that weighs the most.
Symbol: I
Atomic mass: 126.90447 u
Atomic number: 53
Electron configuration: [Kr] 4d105s25p5
(Hope it helps)
how does the internal energy of the ice and water mixture change if the temperature does not rise while the ice is melting?
Answers
Thermal energy is transferred to ice causing this to occur. Since the temperature doesn't change, all the thermal energy is used to increase the potential energy.
What is potential energy?
As we know, an item can shop power because of its function. In the case of a bow and an arrow, when the bow is drawn, it stores some amount of electricity, which is chargeable for the kinetic energy it gains when released.Similarly, within the case of a spring, while it is displaced from its equilibrium position, it profits a few amount of power which we have a look at inside the shape of pressure we sense in our hands upon stretching it. We are able to outline capacity power as a shape of power that outcomes from the alteration of its position or kingdom.To know more about potential energy, click the link given below:
https://brainly.com/question/24284560
#SPJ4
Is the one piece real?
Answers
Answer:
Yes
Explanation:
The one piece, the one piece is real.
What are some of the main reasons calcium chloride can be beneficial Why can it be useful?
Answers
Because calcium chloride has a lower freezing point than sodium chloride, it is more efficient at melting the ice on roadways in the winter.
Because the calcium chloride solution has a lower freezing point when combined with water than the sodium chloride solution, using calcium chloride solution on roadways in the us is easier.
The van't hoff factor causes the calcium chloride solution to have a lower freezing point.
Since calcium chloride has a lower freezing point than ice, when we apply it to the ice, it takes heat from the ice and melts it. There is no more efficient way to do the same objective than using sodium chloride solutions.
To know more about Calcium chloride, visit,
https://brainly.com/question/12216405
#SPJ4
Which conditions would activate the necessary enzymes for the citric acid cycle?a. high levels of ATP b. low levels of ADP c. high levels of ADP d. high levels of NADH
Answers
High levels of ATP would activate the necessary enzymes for the citric acid.
The TCA cycle metabolizes acetate that is derived from carbohydrates, proteins, and fats to form adenosine triphosphate (ATP), which is referred as the body's energy currency. Oxidation of one glucose molecule through the combined action of the TCA cycle, glycolysis, and oxidative phosphorylation yields an estimated number of 30–38 ATP molecules.
High levels of ATP activates citric acid cycle because citrate synthase is inhibited by the final product of the citric acid cycle as ATP, ADP (adenosine diphosphate) works as an allosteric activator of the enzyme as ATP is formed from ADP. Therefore, the rate of the citric acid cycle is reduced when the cell has a high level of ATP.
Learn more about TCA cycle from the link given below.
https://brainly.com/question/14845050
#SPJ4
draw the structure of the carbocation intermediate that leads to the indicated products in this reaction.
Answers
carb is the structure of the carbocation intermediate that leads to the indicated products.
Carbocation: It is an organic molecule, an intermediate, that has a carbon atom bearing a positive charge and three bonds instead of four
The reaction given in the question is shown below.
carb1-----fig(1)
Given reaction
The reaction starts with the removal of the bromide ion to generate the carbocation. The carbocation is generated near the four-membered ring.
The methylene shift occurs to rearrange the carbocation by forming a five-membered ring.
carb2----fig(2)
Bromide ion removal
This carbocation accounts for the formation of the products given in the question. Two alkenes are formed by the elimination reaction and the ether is formed by the substitution reaction.
carb3----fig(3)
Elimination and substitution of products
Thus, the carbocation formed in the reaction is shown below.
carb1-------fig(4)
Fig (4) shows the final Structure of the carbocation intermediate.
To know more about carbocation intermediate:
https://brainly.com/question/18503359
#SPJ4
what would happen to your calculated specific heat of the unknown metal if you had failed to completely dry the metal unknown between trials?
Answers
The specific heat would be less than the real number, while the total heat absorbed would be higher.
The amount of energy required to raise the temperature of one gram of a given substance by one degree Celsius is known as the specific heat capacity. All compounds have a unique value that can be used to identify an unknown compound when other methods of identification might not be as useful. One technique, for instance, that may be utilized (with the help of lander robots) to determine the composition of extraterrestrial worlds like Mars is the measurement of specific heat. Additionally, a material's potential industrial applications may be influenced by its specific heat. Materials with low specific heat are frequently employed in heat transmission applications like cooking pots and radiators, while materials with high specific heat can be utilized as insulators in freezers, ovens, etc.
To learn more about specific heat, tap here:
brainly.com/question/11297584
#SPJ4
how many equivalent resonance structures can you draw for sulfate where the formal charges a minimized (like in the previous question)?
Answers
There isn't a most favorable equivalent resonance of the Sulfate ion because they are all identical in charge and there is no change in Electronegativity between the Oxygen atoms.
What is atom?
An atom is a particle of count that uniquely defines a chemical element. Atoms include a critical nucleus surrounded with the aid of using one or greater negatively charged electrons. The nucleus is undoubtedly charged and carries one or greater quite heavy debris referred to as protons and neutrons.
Therefore, There isn't a most favorable equivalent resonance of the Sulfate ion because they are all identical in charge and there is no change in Electronegativity between the Oxygen atoms.
To learn more about atom refer the given link:-
https://brainly.com/question/16001188
#SPJ4
If 19.9% of Boron is Boron-10 and 80.1% is Boron-11, what is the average atomic mass?
Answers
Answer:
10.801=11
Explanation:
avarage atomic mass =relative abundance of first isotope*mass number of first isotope/100+relative abundance of second isotope*mass number of second isotope/100
The melting point of gold is 3,533 ºF, while the boiling point is 9,343 ºF. What phase is gold in when the temperature is 5,262 °C?
Answers
ANSWER:
the melting point of gold is approximately 1,946 ºC and its boiling point is approximately 5,195 ºC. Therefore, if the temperature is 5,262 ºC, gold would be in its liquid phase. It is worth noting that the temperature you provided is not in the correct units, as it should be in degrees Celsius (ºC) instead of degrees Fahrenheit (ºF). To convert a temperature from Fahrenheit to Celsius, you can use the following formula:
Celsius = (Fahrenheit - 32) * (5/9)
For example, to convert the melting point of gold from Fahrenheit to Celsius, you would do the following:
Melting point in Celsius = (3,533 - 32) * (5/9) = 1,946 ºC
Similarly, you can convert the boiling point of gold from Fahrenheit to Celsius using the same formula:
Boiling point in Celsius = (9,343 - 32) * (5/9) = 5,195 ºC
I hope this helps! Let me know if you have any other questions.
a sample of gas weighs 3.33 g and occupies a volume of 1.365 l at 95 °c and 790 torr. identify the gas sample.
Answers
Given that 3.33 g of the gas, at 95 °C and 790 torr, occupy a volume of 1.365 L, it is chlorine gas (Cl2).
We will start by counting the amount of moles of the gas. This can be done as follows:
(V) = 1.365 L,
(T) = 95 °C,
(P) = 790 torr,
(n) =?
(T) = 95 + 273 = 368 K
(P) = 790 / 760 = 1.039 atm
(R) = 0.0821 atm
PV = nRT
1.039 * 1.365 = n * 0.0821 * 368
n = 0.0469 mole L/Kmol
Finally, we will ascertain the gas's identity as follows:
n = 0.0469 mole
M = 3.33 g
Molecular mass =?
Molar mass is equal to the product of the mass and the mole.
Molar mass equals 3.33 minus 0.0469
Gas molar mass = 71 g/mol
As a result, we can infer that the gas is chlorine gas (Cl2) because
(Cl) = 35.5 g/mol.
(Cl2) = 2 * 35.5 g/mol
= 71 g/mol
To know more about identity of gas, visit:
https://brainly.com/question/16016861
#SPJ4
If the bond angle between two adjacent hybrid orbitals is 120°, which is the hybridization?
a. sp3d2
b. sp
c. sp3
d. sp2
Answers
Option (d). If the bond angle between two adjacent hybrid orbitals is 120 degree then this involves SP2 hybridization.
SP2 hybridization is the mixing of one s and two p atomic orbitals which involves the promotion of one electron in the s orbital to one of the 2p atomic orbitals. The combination of these atomic orbitals creates three new hybrid orbitals equal in energy-level. The hybrid orbitals are higher in energy than the s orbital but lower in energy than the p orbitals, but they are closer in energy to the p orbitals. The new set of formed hybrid orbitals creates trigonal structures creating a molecular geometry of 120 degrees. When the hybridization occurs the molecules have a linear arrangement of the atoms with a certain bond angle. A bond angle is the angle between any two bonds that include a common atom, usually measured in degrees.
To learn more about Hybridization please visit:
https://brainly.com/question/22765530
#SPJ4
2. The melting point of tin is about 232°C. Germanium is an element in the same group as tin on the periodic table, but its melting point is about 938°C. What could you infer about the metallic bonding in Germanium from this information?
Answers
Since the melting point of Germanium is higher than Tin, the metallic bonding in Germanium is stronger than that of Tin.
What is metallic bonding?Metallic bonding is the bonding that exists between the atoms of the elements in a metal.
Metallic bonding occurs as a result of the electrostatic attraction between an electron cloud of delocalized electrons and the positively charged metal ions.
The stronger the electrostatic forces of attraction between the electron cloud of delocalized electrons and the positively charged metal ions, the stronger will be the metal and the higher will be the melting point of the metallic atom.
The melting point of a solid is the temperature at which the solid is converted to heat on the addition of heat without an increase in the temperature of the solid.
Learn more about metallic bonding at: https://brainly.com/question/11490390
#SPJ1
calculate the cell potential for the following reaction that takes place in an electrochemical cell at 25°c.
Answers
The Nernst equation can be used to calculate the potential of an electrochemical cell when not under standard conditions. It is also very useful to show how ion concentration affects cell potential and the cell potential is 0.09314 V.
At 298 K, the Nernst equation simplifies to - 0.0592 v protocol Q where Q is the reaction quotient and n is the coefficient of electrons transferred if the redox equations are balanced.
In this cell copper is reduced at the cathode and oxidized.
0.00V. However, the anode C.When 2 +] = 0.0032 M.including cathode [C. When 2 +] = 4.48 M. , so the cell potential is not 0.00 V. The cell potential can be solved using the Nernst equation. where n = 2 and Q = 0.0032/ 4.48.= 0.0592.E= 0-0.0592/ 2 log (0.000174)E= 0-0.0592/ 2 -3.14E= 0-0.0592/ 2E = 0.09314 V.Read more about potential;
https://brainly.com/question/24142403
#SPJ4
What are the 3 major types of chemical bonds?
Answers
1. Covalent- Nonmetal & Nonmetal
2. Ionic- Metal & Nonmetal
3. Metallic- Metal & Metal
Please give brainliest
A sealed cylinder fitted with a movable piston contains ideal gas at 27°c, pressure 0.500 × 10^5 pa, and volume 1.1 m^3. What will be the final temperature if the gas is compressed to 0.800 m3 and the pressure rises to 0.820 × 10^5 pa?
Answers
Using the combination law equation, its final temperature would be 42°C B K if the gas was compressed to 0.800 m3 and the pressure increased to 0.820105 Pa.
How can the temperature of such a mixture be determined?Apply the equation, where m1 и m2 are the weights of water contained in the first and two containers, T1 and T2 are the temperatures of the water contained in the first and second containers, to determine the final temperature mixture.
T2=0.820×105 Pa0.5×105 Pa0.8 m31.25 m3(300 K =314.88 K
Convert the unit of the temperature from the kelvin to degree Celsius.
T2=314.88 K-273 K =41.88 °C
Are the starting temperature and the ending temperature the same?It is necessary for the final temperature to match the starting temperature. The internal energy must match the internal energy at the beginning and end. There must be no net heat added to the system during the procedure.
To know more about final temperature visit:
https://brainly.com/question/11244611
#SPJ4
When a nucleus of 235U undergoes fission, it breaks into two smaller, more tightly bound fragments. Calculate the binding energy per nucleon for 235U and for the fission product 137Cs .
Answers
The binding energy per nucleon for 235U and 137Cs is 7.6 MeV and 8.39 MeV.
The minimal amount of energy needed to take a particle out of a system of particles or to break a system of particles down into its component pieces is known as binding energy (BE).
Given nucleus = 235 U and nucleon = 137 Cs.
a) The expression to find BE of 235 U is written as 235U92.
Atomic number (Z) = 92 = number of protons
Mass number (A) = 235 (number of protons + neutrons)
Number of electrons = 235-92 = 143.
Mass of neutron (mₙ) = 1.00866 u
Mass of proton ([tex]m_H[/tex]) = 1.00783 u
The binding energy for the nucleus is calculated using the below formula,
[tex]B = (Zm_H + Nm_n- m_{atom})\times\mathrm{931.49\;MeV/u}[/tex]
For 235U,
[tex]\begin{aligned}BE&=[(92\times1.00783 u)+(143\times1.00866 u)-235](\mathrm{931.49\;MeV/u})\\&=\mathrm{1783.8\;MeV}\end{aligned}[/tex]
The BE per nucleon is given by,
[tex]\begin{aligned}\text{BE per nucleon}&=\frac{\text{total BE of nucleus}}{\text{number of nucleon}}\\&=\frac{1783.8}{235}\\&=\mathrm{7.6\;MeV}\end{aligned}[/tex]
b) The expression to find BE of 137Cs is written as 137Cs55.
Atomic number (Z) = 55 = number of protons
Mass number (A) = 137 (number of protons + neutrons)
Number of electrons = 137-55 = 82.
Mass of neutron (mₙ) = 1.00866 u
Mass of proton ([tex]m_H[/tex]) = 1.00783 u
The atomic mass of Cs = 137u
For 137Cs,
[tex]\begin{aligned}BE&=[(55\times1.00783 u)+(82\times1.00866 u)-137](\mathrm{931.49\;MeV/u})\\&=\mathrm{1149.3\;MeV}\end{aligned}[/tex]
The BE per nucleon is given by,
[tex]\begin{aligned}\text{BE per nucleon}&=\frac{\text{total BE of nucleus}}{\text{number of nucleon}}\\&=\frac{1149.3}{137}\\&=\mathrm{8.39\;MeV}\end{aligned}[/tex]
The answers are 7.6 MeV and 8.39 MeV.
To know more about binding energy:
https://brainly.com/question/10095561
#SPJ4
Conduction chocolate candy in your hand will eventually melt as the heat from your body is released and makes it melted.
Answers
Conduction causes the Thermal energy flows out of your hand into the chocolate bar and it starts off evolved to soften.
This approach that your (hand) is shifting strength to the chocolate bar inflicting it to revel in a segment extrade from stable to liquid. Conduction Chocolate sweet for your hand will finally soften as the warmth out of your frame is launched and makes it melted.
The melting factor of chocolate falls among 86°F and 90°F. This is substantially decrease than the common temperature of the human frame, that's 98.6°F so the warmth out of your hand increases the temperature of the chocolate and reasons it to soften.
Read more about the heat:
https://brainly.com/question/934320
#SPJ4
a sample of neon gas collected at stp occupies a volume of 37.3 l. how many moles of gas does the sample contain?
Answers
2.17 moles of gas does the sample contain in a sample of neon gas collected at stp occupies a volume of 37.3 l.
What is noble gas?
Noble gases form a class of chemical elements with similar properties. Under standard conditions, they are all odorless, colorless, monatomic gases with little chemical reactivity. The six naturally occurring noble gases are helium, neon, argon, krypton, xenon, and radioactive radon.
Therefore, 2.17 moles of gas does the sample contain in a sample of neon gas collected at stp occupies a volume of 37.3 l. Noble gases form a class of chemical elements with similar properties. Under standard conditions, they are all odorless, colorless, monatomic gases with little chemical reactivity.
To learn more about noble gas refer the given link:-
https://brainly.com/question/11414033
#SPJ4
What is the composition of the sigma bonds in methane i.e. what atomic orbits comprise the molecular orbit? Hint: they are all the same, so there is only one kind sigma bond here.
Answers
Covalent bonds of sigma type are present in methane where in the hybridization of each bond is σ sp³-σ sp³.
What are covalent bonds?Covalent bond is defined as a type of bond which is formed by the mutual sharing of electrons to form electron pairs between the two atoms.These electron pairs are called as bonding pairs or shared pair of electrons.
Due to the sharing of valence electrons , the atoms are able to achieve a stable electronic configuration . Covalent bonding involves many types of interactions like σ bonding,π bonding ,metal-to-metal bonding ,etc.
Sigma bonds are the strongest covalent bonds while the pi bonds are weaker covalent bonds .Covalent bonds are affected by electronegativities of the atoms present in the molecules.Compounds having covalent bonds have lower melting points as compared to those with ionic bonds.
Learn more about covalent bond,here:
https://brainly.com/question/19382448
#SPJ1
Which choice best describes the properties of potassium (k), based on its position on the periodic table of elements?.
Answers
The option that best describes the properties of the element potassium (K), based on its position on the periodic table of chemical elements is that it is a highly reactive metal. Hence, the correct option is B.
Alkali metals can be described as the monovalent chemical elements that are found in Group IA of the modern periodic table.
Generally, chemical elements of alkali metals readily lose their single (one) valence electron to form ionic compounds with non-metals. Some examples of alkali metal are given below,
Lithium (L).
Sodium (Na).
Potassium (K).
Potassium (K) is an alkali metal with a single (one) valence electron in its outermost shell and is considered to be highly reactive with other chemical elements, especially halogens because of its position on the modern periodic table of chemical elements.
We can conclude that, Potassium (K) is a highly reactive metal based on its position on the periodic table of chemical elements.
Learn more about alkali metals from the link given below.
https://brainly.com/question/20664156
#SPJ4
Three students were discussing their ideas about chemical bonds. This is what they said:
Janre: "I think a chemical bond is produced by a molecule. It is a substance made up of matter that holds atom together."
Will: "I think a chemical bond is an attraction between atoms. It is not made up of matter."
Leta: "I think a chemical bond is a structural part of an atom that connects to other atoms."
Which student do you agree with and why? Explain your thinking.
Answers
Answer:
I agree with Janre. A chemical bond is produced by a molecule, which is a substance made up of matter that holds atoms together. A chemical bond is the result of the attraction between atoms, which allows them to combine and form molecules. This attraction is not made up of matter itself, but it is a fundamental property of atoms that allows them to form bonds and create molecules. Therefore, Janre's explanation is the most accurate and complete.
Explanation:
Answer: In this case, I find myself in agreement with Janre. A molecule is a substance made up of matter that binds atoms together via a chemical bond. When atoms come together due to mutual attraction, a chemical bond is formed. This attraction is not material, but rather a feature of atoms that is essential to the formation of bonds and molecules. Janre's explanation is the most thorough and precise one available.
Explanation:
According to molecular orbital theory, the regions of the wave function with the highest probability of finding electrons are areas with _______.
Answers
According to molecular orbital theory, the regions of the wave function with highest probability of finding electrons are areas with constructive interface.
Molecular orbital theory describes the behavior of electrons in a molecule in terms of combinations of the atomic wavefunctions. The resulting molecular orbitals may extend over all the atoms in the molecule. Bonding molecular orbitals are formed by in-phase combinations of atomic wavefunctions, and electrons in these orbitals stabilize a molecule. Antibonding molecular orbitals result from out-of-phase combinations and electrons in these orbitals make a molecule less stable. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. Molecular orbitals are combinations of atomic orbital wave functions.
To learn more about Molecular Orbital Theory please visit:
https://brainly.com/question/17371976
#SPJ4
dding a small amount of strong acid to a buffer solution will cause the ph to increase slightly. true false
Answers
True, The pH of a buffer solution will be affected by strong acid because strong acid dissociates completely to give H+ ions so the pH of buffer will be appreciably affectedand as the H+ concentration increased pH will decrease.
When a small volume of acid is added to a buffer solution, the pH of the system decreases as an effect of the increased hydronium ions in the solution. However, when a small volume of base is added to a buffer solution, the pH of the system increases as an effect of the increased hydroxide ions in the solution.When a base is added to a buffer solution, the pH does not change. The buffer solution prevents the base from neutralizing the acid.
To know more about Buffer Solution:
brainly.com/question/29802312
#SPJ4
a pure substance which can only be seperated into two or more simpler substances using chemcial changes is called
Answers
A pure substance that can only be separated into two or more simpler substances using chemical changes is called a chemical compound.
A chemical compound is a type of pure substance that is made up of two or more different elements that are chemically bonded together. These elements are combined in a fixed ratio, and the resulting compound has different properties than the individual elements. For example, water (H2O) is a chemical compound made up of two hydrogen atoms and one oxygen atom. The atoms are bonded together by covalent bonds, and the resulting compound has the unique properties of being a liquid at room temperature and being essential for life.In order to separate a chemical compound into its constituent elements, a chemical reaction is required. This process involves breaking the chemical bonds between the atoms and forming new bonds between the atoms of the individual elements. This can be done through a variety of methods, such as electrolysis or heating the compound to high temperatures. The resulting elements will have the same physical and chemical properties as the original elements, but will no longer be bonded together as a compound.
to know more about element-
https://brainly.com/question/28948712
#SPJ4