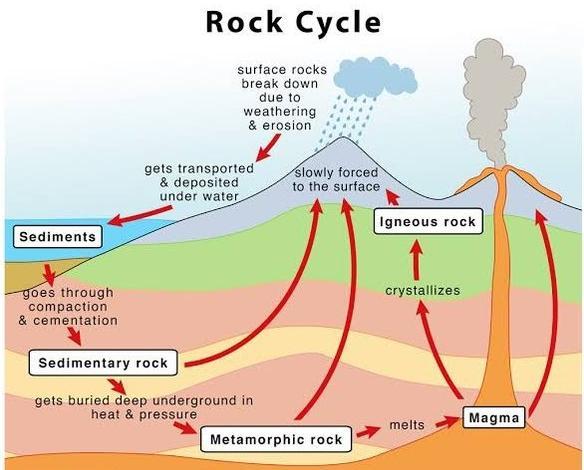
Create a model using images that would show what would happen to the igneous rock when it is exposed to different energy sources.
Answers
igneous is moved to Earth's surface and exposed to energy from the sun, it could weather into smaller rock pieces that could form sedimentary rock

Related Questions
what is the maximum number of hydrogen ions that can be removed by a base from carbonic acid h2co3? one two three or more g
Answers
Answer: Both the hydrogen atoms present in carbonic acid are replaceable and thus the maximum no of H atoms can be removed is 2.
Explanation:
Hydrogen is not bonded to another hydrogen in carbonic acid. Although the shorthand formula is H2CO3 which must be the reason behind the question, the structure is actually:
Meaning that each of the hydrogen atoms is in resonance with an oxygen. Since the oxygens are by themselves very electronegative, meaning they attract the hydrogen electron very strongly and stably, they can easily loose the hydrogen proton.
Learn more about H2CO3 here https://brainly.com/question/19052059
#SPJ4
HELP HELP PLEASE ASAP
Answers
Answer:
1.)3.33045
2.)0.02550535714
3.)53.21
4.)26.56
5.)6.72672
6.)3.351
7.)843750
8.)227376.25
9.)0.00172776
10.)8.9577346e+33
Explanation:
What is the correct electron configuration for Si atomic number
O A. 1s22s22p 3s23p²
OB. 1s22s22p 3s²3p³
O C. 1s22s22p63s²2d³
O D. 1s22s22p63s¹3p³
Answers
1s22s22p63s^13p^3 is correct electron configuration for SI atomic number.
What is electron configuration?
The distribution of electrons in an element's atomic orbitals is described by the element's electron configuration. Atomic electron configurations adhere to a standard notation in which all atomic subshells that contain electrons are arranged in a sequence with the number of electrons they each hold written in superscript. For instance, sodium's electron configuration is 1s22s22p63s1. Long electron configurations are frequently produced by the standard notation. In these circumstances, a shortened or condensed version of the standard notation may be used. The sequence of fully filled subshells that represent the electronic configuration of a noble gas are replaced in the abbreviated notation by the symbol for that noble gas enclosed in square brackets.
To know more about configuration click below
https://brainly.com/question/26084288
#SPJ1
Silicon dioxide reacts with carbon to form silicon carbide and carbon monoxide as shown in the unbalanced reaction below. Determine the percent yield of silicon carbide if 79.1 grams of carbon reacts with an excess of silicon dioxide and 67.4 g of silicon carbide is actually recovered.
SiO2 + C → SiC + CO. I’m working on this problem and I can not figure out where they got the 3 from
Answers
The percent yield of SiC is 76.6% if 79.1 grams of carbon reacts with an excess of silicon dioxide and 67.4 g of silicon carbide is actually recovered.
The first step is to balance the equation that is given. On balancing we get,
SiO2 + 3C → SiC + 2CO
In the Next step, we need to Determine the theoretical amount of silicon carbide produced (if all carbon reacted).
"Excess silicon dioxide" in the question can be interpreted that carbon being the limiting reactant, and thus the amount of silicon carbide produced depends on how much carbon is available to react in the reaction.
From the balanced equation, it could be understood that for every 3 moles of carbon reacted, 1-mole silicon carbide is produced. Therefore,
Molar mass carbon = 12.01 g/mol
Molar mass silicon carbide = 28.09 g + 12.01 g
Molar mass silicon carbide = 40.1 g/mol
Then,
Theoretical yield = 79.1 g x (1 mol / 12.01 g) x (1 mol / 3 mol) x (40.1 g / 1 mol)
Theoretical yield = 88.04 g SiC
Therefore the theoretical yield is 88.04g
From the given values the percent yield can be computed as,
Percent yield = (actual yield of SiC / theoretical yield of SiC) x 100%
= (67.4 / 88.04) x 100%
= 0.766 x 100%
% yield of SiC = 76.6%
Therefore, the percent yield of SiC is 76.6%
To know more about percent mass, click below:
https://brainly.com/question/26150306
#SPJ1
pls answer question will mark brainliset tyty
Answers
Answer:
Graph D.
Explanation:
A certain polyatomic ion contains 49 protons and 50 electrons. What's the net charge of this ion?Question options:A) +1B) +2C) –1D) –2
Answers
Explanation:
We have to find the net charge of a polyatomic ion that contains 49 protons and 50 electrons.
n° of protons = 49 n° of electrons = 50
The charge of each electron is -1 and the charge of each proton is +1.
charge of electron = -1 charge of proton = +1
Finally the net charge will be:
Net charge = n° of electrons * charge of electron + n° of protons * charge of proton
Net charge = 50 * (-1) + 49 * (+1)
Net charge = -1
Answer: C) -1
6. You have 400 mL of a 6 M solution of Ca(NO3)2. How many grams are present?
Answers
We are given the liters and molarity of the solution. To find grams we must take into account the definition of molarity. Molarity is defined as:
[tex]Molarity=\frac{MolesSolute}{Lsolution}[/tex]Now, we find the moles of solute:
[tex]\begin{gathered} MolesSolute=Molarity\times Lsolution \\ MolesSolute=6M\times400mL\times\frac{1L}{1000mL} \\ MolesSolute=6\frac{mol}{L}\times0.400L=2.4mol \end{gathered}[/tex]Now, to convert these moles to grams we must multiply the moles by the molar mass of Ca(NO3)2. Molar mass Ca(NO3)2 is 164.10g/mol.
So, the grams will be:
[tex]\begin{gathered} gCa(NO_3)_2=givenmolCa(NO_3)_2\times\frac{MolarMass,gCa(NO_3)_2}{1molCa(NO_3)_2} \\ gCa(NO_3)_2=2.4molCa(NO_3)_2\times\frac{164.10gCa(NO_3)_2}{1molCa(NO_3)_2} \\ gCa(NO_3)_2=394gCa(NO_3)_2 \end{gathered}[/tex]Answer: There are present 394grams of Ca(NO3)2
In clinical applications, the unit parts per million (ppm) is used to express very small concentrations of solute, where 1 ppm is equivalent to 1 mg of solute per 1 L of solution. Calculate the concentration in parts per million for each of the solutions.There is 43 μg of calcium in a total volume of 87 mL .concentration of calcium: _________ppm There is 0.91 mg of caffeine in a total volume of 105 mL .concentration of caffeine: _________ppm There is 0.47 mg of trace particles in a total volume of 95 dL .concentration of trace particles: ___________
Answers
Answer & Procedure
To solve this problem you will need to transform all the concentrations to the equivalence of ppm, which is 1 mg of solute per 1 L of solution.
Calcium: There are 43 μg of calcium in a total volume of 87 mL.
[tex]\frac{43\mu g}{87\text{ mL}}\frac{1\text{ mg}}{1000\text{ }\mu g}\frac{1000\text{ mL}}{1\text{ L}}=0.49\frac{mg}{L}=0.49\text{ ppm}[/tex]Caffeine: There is 0.91 mg of caffeine in a total volume of 105 mL.
[tex]\frac{0.91\text{ mg}}{105\text{ mL}}\frac{1000\text{ mL}}{1\text{ L}}=8.67\frac{mg}{L}=\text{ 8.67 ppm}[/tex]Trace particles: There is 0.47 mg of trace particles in a total volume of 95 dL.
[tex]\frac{0.47\text{ mg}}{95\text{ dL}}\frac{10\text{ dL}}{1\text{ L}}=0.049\frac{mg}{L}=0.049\text{ ppm}[/tex]Indicate whether each of the following chemical conversions requires an oxidizing agent (o), a reducing agent (r), or neither (n).
Answers
An oxidizing agent is required. In c), there's no extrude withinside the oxidation numbers, so nor is required. Indicate whether or not every of the subsequent chemical conversions REQUIRES an oxidizing agent (O), a decreasing agent (R), or neither (N).
a) NO31- NH3
b) PO33- PO43-
c) AgCl Ag1+ + Cl1-
a) NO3- --> NH3 Requires a decreasing agent
b) PO33- --> PO43- Requires an oxidizing agent
c. AgCl --> Ag+ + Cl- There isn't any extrude in oxidation wide variety.
You discern this out with the aid of using searching on the oxidation numbers of factors withinside the equations. In the primary one, the oxidation wide variety of N decreases. Since that is a reduction, a decreasing agent is required. In b), the oxidation wide variety of P increases. Since that is an oxidation, an oxidizing agent is required. In c), there's no extrude withinside the oxidation numbers, so nor is required.
Learn more about oxidizing agent here https://brainly.com/question/1390694
#SPJ4
The frequency of a wave is 2.8 x 10^3 Hertz. What is the wavelength of these gamma rays?
Show all work!
Answers
The frequency of a wave 2.8 x 10³ Hertz is then the wavelength of these gamma rays is 107 nm
Wavelength is the the distance between two successive crests or troughs of a wave
Here given data is
Frequency = 2.8 x 10³ Hertz
Velocity of gamma rays = 3×10⁸m/s
We have to calculate wavelength of these gamma rays?
So, wavelength = velocity/frequency
λ = v/f
λ = 3×10⁸m/s/2.8 x 10³ Hertz
λ = 107 nm
Know more about wavelength
https://brainly.com/question/11483057
#SPJ1
ASAP
Compare the heat energy in a teaspoon of boiling water and a swimming pool full of room temperature water.
This is an essay question
Answers
The heat energy teaspoon of boiling water and a swimming pool full of room temperature water then swimming pool have more heat energy than the teaspoon of boiling water
Heat energy is the result of movement of tiny particles called as atom, molecule, or ion in solid liquid and gases and heat energy is the transfer from one object to the another and in Celsius scale water freezes at 0°C and boil at 100°C to 90°C however the swimming pool contains a lot more water therefore the pool has more thermal energy than the cup of tea even though the tea is hotter than the water in the pool that's why heat energy is more in the swimming pool then in teaspoon of boiling water
Know more about heat energy
https://brainly.com/question/29210982
#SPJ1
Explain the Different types of electromagnetic wave
Answers
Answer:
Seven waves make up the electromagnetic spectrum.
Explanation:
Radio Waves
Used in television and radio broadcasts
Microwaves
Used for satellite communications and heating up food
Infrared radiations
Used in electric heaters and other heating devices.
Visible light
used for fibre optic communications
Ultraviolet rays
used in energy-efficient lamps, sun tanning
X-rays
Medical imaging and treatments
Gamma rays
used for sterilizing and disinfecting
Wavelengths down the spectrum increase as the frequency decreases.
How many joules of heat are needed to completely vaporize 24.40 grams of water at its boiling point?Given ΔHvap = 40.6 kJ/mol
Answers
55,035.5 Joules
Explanations:Heat of vaporisation is the amount of energy required to transform a mole of liquid water into gas at constant temperature.
Given the following parameters:
Mass of water = 24.40 grams
ΔHvap = 40.6 kJ/mol
Determine the moles of water
Mole = mass/molar mass
Mole = 24.40/18
Mole of water = 1.356moles
Determine the required heat required
Q = mole of water * ΔHvap
Q = 1.356moles * 40.6kJ/mole
Q = 55.0355kJ
Q = 55.0355 * 1000
Q = 55,035.5 Joules
Hence joules of heat needed to completely vaporise 24.40 grams of water at its boiling point is 55,035.5 Joules
Do you think the substances after the reaction was still copper (II) chloride and aluminum?
Answers
When aluminum is reacts copper (II) chloride then it will form aluminum chloride and Copper.
Aluminum as well as copper(II) chloride combine very vigorously, causing the reaction mixture to become extremely hot as heat was produced, the aluminum foil to breakdown, a reddish brown solid to appear, as well as gas bubbles to be released.
The chemical reaction can be written as:
2Al + 3CuCl2 → 3Cu + 2AlCl3
Therefore, Copper and aluminum chloride will be formed after the reaction as a product.
To know more about aluminum
https://brainly.com/question/28488595
#SPJ1
Which one of these statements is usually true about waves?
A.All kinds of waves are the same size.
B.All waves move at the same speed.
C.Waves don't move matter, just energy.
D.Some waves do not move in a pattern.
Answers
Answer: C. Waves don't move matter, just energy.
Explanation:
a 100.0 ml sample of 0.18 m hclo4 is titrated with 0.27 m lioh. determine the ph of the solution after the addition of 50.0 ml of lioh.
Answers
The pH of the solution after addition of 50.0ml is 0.1003
pH is known as the negative logarithm of the hydrogen ion concentration in a solution.
In the given condition we have 100 ml of sample of 0.18 molar of HclO4 titrated with 0.27m LioH.
pH = log[H+]
= log [Hclo4]
Mole of Hclo4 0.1 L * 0.18 m = 0.018 M
mol LiOH = 0.1 L * 0.27 m = 0.027 M
total volume = 0.1L + 0.1L = 0.2L
HClO4] = (0.018-0.0081) /0.2 L
= -1.26 billion
PH = -㏒ -1.26
= 0.1003
So, the pH of the solution after adding 100 ml of liquid is 0.1003
An ionic bond is the result of....Group of answer choicesElectron sharingNon-metals bonded togethercharge attractionMolecules
Answers
An ionic bond is also called an electrovalent bond. It is formed from the electrostatic attraction between oppositely charged ions in a chemical compound.
Answer: charge attraction.
Power is an object's change in _______ over a specific time.
Answers
Power is an objects' change in energy over a specific time.
What is power?We define the term power as the rate of the change of energy with time. We know that the energy of a body has to do with the ability of the body to do work.
This implies that we can be able to write the formula of the power of a body as the following;
Power = Energy/ Time
Thus we can now see that power is the change in energy over a specific time. This is how we can be able to obtain the /power of an object under study.
Learn more about power:https://brainly.com/question/287674
#SPJ1
Describe how a state function depends on the pathway between the initial and final states of a system.
Answers
A state function only depends on the thermodynamic system's starting and current states.
What exactly is a "thermodynamic system"?A thermodynamic system is a collection of objects or a significant region of space. The surroundings or mass outside the system is referred to as such, and the border is the region separating the two.
For instance, if we are examining a car engine, the thermodynamic system is the gasoline burning inside the engine's cylinder, and the system's surrounds are the radiator, exhaust system, piston, and outside air. The inner surfaces of the cylinder and piston then form the boundary.
The cosmos is made up of the system and its surroundings. Thermodynamic systems can be classified as open, closed, or isolated. With its surroundings, an open system can interchange both matter and energy. Since heat and water vapor can be lost to the air, the stovetop example would be considered an open system.
To learn more about thermodynamic system refer to
https://brainly.com/question/27386894
#SPJ4
which of the following is true about cations?a. They are negatively charged and move toward an anodeb. They are negatively charged and move toward a cathodec. They are positively charged and move toward an anoded. They are positively charged and move toward an cathode.
Answers
ANSWER
They are positively charged and move toward an anode
Option C
EXPLANATION
In electrochemical reaction, cation are positively charged. Since they are positively charged, they move toward negatively charged anode
There are two types of reaction which are oxidation and reduction reaction. At oxidation, the positively charged cations move towards the negatively charged anode.
Recall, that like charges attract and unlike charges repel.
Therefore, the correct answer is option C
3.25 kcal is the same amount of energy as A. 3.25J. B. 0.7771. C. 777J. D. 13600 j.
Answers
Kilocalorie is a unit of measuring the amount of energy of a reaction, but this is not the only unit, we can also have Joules as a unit, and the conversion is:
1 Kcal = 4184 Joules
Therefore if we have 3.25 Kcal, we will have:
3.25 * 4184 = 13600 Joules of energy, therefore letter D
What type of mechanical mixture is pop
Answers
Hope this helped!
Help a girl out I’m rushing
Answers
Answer:
spring
Explanation:
What amino acid sequence is encoded by the codon sequence GCCGCGACCGCUACU?
Answers
Answer
Ala-Ala-Thr-Ala-Thr
Procedure
We will use the genetic code in the table below to solve this question.
Every three consecutive letters need to be matched with the given code
A good strategy s to split the sequence as follows: GCC-GCG-ACC-GCU-ACU
After matching, we have:
Ala-Ala-Thr-Ala-Thr
According to the beer-lambert law, what happens to the absorbance value of a light-absorbing chemical when its concentration increases?.
Answers
According to the beer-lambert law when its concentration increases so does the absorbance increase.
beer-lambert law states that the amount of energy absorbed or transferred by a solution is proportional to the molar absorptivity of the solution and the concentration of the solute.
This means that concentrated solutions absorb more light than dilute solutions.
so the According to the beer-lambert law concentration increases, so does the absorbance. Therefore, absorbance is directly proportional to concentration.
learn more about beer-lambert law at https://brainly.com/question/8831959
#SPJ4
2. You were asked to examine a prepared slide under the HPO. Your eyepiece is 20x and the HPO is 80x. How many times was the specimen magnified?
Answers
Answer:
Explanation:
Magnification = 20 * 80
1600
In a solution with a pH of 3 a. Litmus Is blue and phenolphthalein is colorless b. Litmus is blue phenolphthalein is red c. Litmus Is red phenolphthalein is colorless d. Litmus is red phenolphthalein is red.
Answers
Answer
c. Litmus Is red phenolphthalein is colorless
Explanation
A solution with a pH of 3 is acidic. The main use of litmus is to test whether a solution is acidic or basic, blue litmus turns red under acidic conditions, and red litmus turns blue under basic or alkaline conditions. Phenolphthalein is often used as an indicator in acid–base titrations. phenolphthalein, it turns colorless in acidic solutions and pink in basic solutions.
Therefore, the correct option for a solution with a pH of 3 is:
c. Litmus Is red phenolphthalein is colorless
what will increase the rate of polymerization and decrease the activation energy necessary for further polymerization?
Answers
Catalysts will increase the rate of polymerization and decrease the activation energy necessary for further polymerization.
Polymerization is a chemical reaction that involves the joining of simple molecules to generate long-chain molecules. Catalysts accelerate polymerization by decreasing the activation energy required for further polymerization. Catalysts can be light, heat, water, acids, or other compounds. Uncontrolled polymerization frequently results in a massive release of energy. In the blue and yellow portions of the ERG, materials that may undergo violent polymerization if exposed to heat or pollution are denoted with a P.
Inhibitors are compounds that are applied to easily polymerizing products to inhibit or avoid an undesirable reaction. Inhibitors increase the amount of activation energy required.
Therefore catalysts will increase the rate of polymerization and decrease the activation energy necessary for further polymerization.
To learn more about polymerization visit
https://brainly.com/question/18701826
#SPJ4
a barometer reads the atmospheric pressure 764.7 torr. a sample of gas is placed in vessel attached to an open-end mercury manometer. the level of mercury in the open-end arm of the monometer has a height of 136.4 mm, and that in the arm that is in contact with the gas has a height 103.8 mm. what is the pressure of gas in atmosphere
Answers
In this question we are given with the value of atmospheric pressure 764.7 torr.
And a sample of gas is placed in vessel attached to an open-end mercury manometer.
The level of mercury in the open-end arm of the monometer has a height of 136.4 mm and that in the arm that is in contact with the gas has a height 103.8 mm.
We know that,
h is the difference of heights of manometer and contact point
h = 136.4 - 103.8
h = 32.6 mm
On considering acceleration to the gravity, g = 10m/ss
Pressure inside the glass = dgh
So,
pressure of gas in atmosphere = 764.7 + 32.6 = 797.3
To know more about Atmospheric Pressure:
brainly.com/question/28310375
#SPJ4
During the process of making hydrogen fuel, what are the byproducts? Are they harmful to humans or the environment?